How many miles are in 200 km? Pick the correct set up below, do not solve....
Fantastic news! We've Found the answer you've been seeking!
Question:


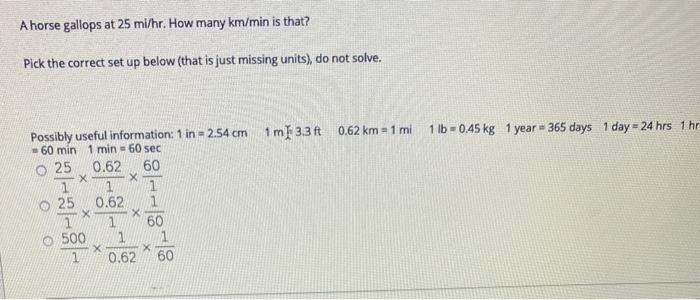
Transcribed Image Text:
How many miles are in 200 km? Pick the correct set up below, do not solve. Possibly useful information: 1 in = 2.54 cm 1 m = 3.3 ft 0.62 km 1 mi O200 km 1 O200 km 1 X X X 200 km 1 1 mi 0.62 km .062 mi 1 km 0.62 km = 1 mi 1 lb = 0.45 kg How many minutes are in 12.5 days? Pick the correct set up below (that is only missing units), do not solve. Possibly useful information: 1 yr = 365 days 1 day = 24 hrs 1 hr = 60 min 1 min = 60 sec O 12.5 24 1 O 12.5 1 -X 1 24 24 O 12.5 -88-8 X 11 X 1 60 1 X 60 60 1 A horse gallops at 25 mi/hr. How many km/min is that? Pick the correct set up below (that is just missing units), do not solve. Possibly useful information: 1 in = 2.54 cm 1m 3.3 ft 0.62 km = 1 mi 60 min 1 min = 60 sec 0.62 60 1 O 25 1 1 25 0.62 1 X X 1 500 X x 1 60 1 1 X X 1 0.62 60 1 lb 0,45 kg 1 year = 365 days 1 day 24 hrs 1 hr How many miles are in 200 km? Pick the correct set up below, do not solve. Possibly useful information: 1 in = 2.54 cm 1 m = 3.3 ft 0.62 km 1 mi O200 km 1 O200 km 1 X X X 200 km 1 1 mi 0.62 km .062 mi 1 km 0.62 km = 1 mi 1 lb = 0.45 kg How many minutes are in 12.5 days? Pick the correct set up below (that is only missing units), do not solve. Possibly useful information: 1 yr = 365 days 1 day = 24 hrs 1 hr = 60 min 1 min = 60 sec O 12.5 24 1 O 12.5 1 -X 1 24 24 O 12.5 -88-8 X 11 X 1 60 1 X 60 60 1 A horse gallops at 25 mi/hr. How many km/min is that? Pick the correct set up below (that is just missing units), do not solve. Possibly useful information: 1 in = 2.54 cm 1m 3.3 ft 0.62 km = 1 mi 60 min 1 min = 60 sec 0.62 60 1 O 25 1 1 25 0.62 1 X X 1 500 X x 1 60 1 1 X X 1 0.62 60 1 lb 0,45 kg 1 year = 365 days 1 day 24 hrs 1 hr
Expert Answer:
Answer rating: 100% (QA)
SOI 1 Given that 2 3 062 km 1 mile 1km 1 mile imi 062 km So Opt... View the full answer

Related Book For 

Quantitative Chemical Analysis
ISBN: 9781319164300
10th Edition
Authors: Daniel C. Harris, Charles A. Lucy
Posted Date:
Students also viewed these chemistry questions
-
How far does a horse travel if it gallops at an average speed of 25 km/h for 30 min?
-
A mass of 5 kg is tied to an elastic cord, 5 m long, and dropped from a tall bridge. Assume the cord, once straight, acts as a spring with k = 100 N/m. Find the velocity of the mass when the cord is...
-
A piston/cylinder contains 2 kg water at 200C, 10 MPa. The piston is slowly moved to expand the water in an isothermal process to a pressure of 200 kPa. Any heat transfer takes place with an ambient...
-
Which of the following is characteristic of a traditional cost system? a. Reliance on financial performance measures b. Many work in process account transactions c. Many process control points d. All...
-
Some games of strategy are cooperative. One example is deciding which side of the road to drive on. It doesn't matter which side it is as long as everyone chooses the same side. Otherwise, everyone...
-
Glenn Grimes is the founder and president of Heartland Construction, a real estate development venture. The business transactions during February while the company was being organized are listed as...
-
The comparative financial statements of Bien Taco Restaurants for 2010, 2009, and 2008 include the following selected data: Requirements 1. Compute these ratios for 2010 and 2009: a. Quick ratio. b....
-
Jake Tweet hosts a psychology talk show on KRAN radio. Jakes advice averages 10 minutes per caller but varies according to an exponential distribution. The average time between calls is 25 minutes,...
-
Logistics Solutions provides order fulfillment services for dot.com merchants. The company maintains warehouses that stock items carried by its dot.com clients. When a client receives an order from a...
-
Hemming Company reported the following current-year purchases and sales for its only product. Date Activities January 11 Beginning inventory. January 10 Sales March 14 March 151 July 30 October 5...
-
Explain, what are the purpose of SUID and SGID . Provide an example of when they may be used.
-
The classification of intangible assets includes goodwill. Current FASB practice states that goodwill cannot be created, added to, or depreciated or amortized. Some companies have increased their...
-
Why is Cash Flow as important as Profit, particularly to a small firm?
-
your grandfather charles is a widower. he received $2024 per month social security for all of 2019. his only other source of income is part time job at the local library. he receives a w-2 for $14100...
-
Juarez Builders incurred $285,000 of labor costs for construction jobs completed during the month of August, of which $212,000 was direct and $73,000 was indirect supervisory costs.What will be the...
-
Refer to the Harris-Todaro migration model. Assume that unemployment is possible and the manufacturing wage is fixed at M1. What is the urban labour pool?
-
Company K recently acquired Company M. Company K now sells its own products and Company M's products together. As a result, Company K's sales force needs to market Company M's products as well. How...
-
1. Using the information from Problem 16-4B, prepare a statement of cash flows for Lim Garden Supplies Inc. using the direct method of presenting cash flows from operating activities. 2. How does Lim...
-
What is the difference between luminescence, chemiluminescence, and bioluminescence?
-
Which column is more efficient: plate height=0.1 mm plate height=0.1 mm or plate height=1 mm? plate height = 1 mm?
-
a. Use Troutons rule, Hvap (88 J mol-1 K-1) H vap (88 J mol -1 K -1 ) to estimate the enthalpy of vaporization of octane (b.p. 126C). (b.p. 126C) b. Use the form of the Clausius-Clapeyron equation...
-
A Cobb-Douglas production function relates production \((Q)\) to factors of production-capital \((K)\), labor \((L)\), and raw materials \((M)\) - and an error term \(u\) using the equation...
-
Equations (7.13) and (7.14) show two formulas for the homoskedasticity-only \(F\)-statistic. Show that the two formulas are equivalent. Equation (7.13) Equation (7.14) F = (SSR restricted - SSR...
-
Suppose a researcher collects data on houses that have sold in a particular neighborhood over the past year and obtains the regression results in the table shown below. a. Using the results in column...

Study smarter with the SolutionInn App


