How many mL of 0.602 M HNO3 are needed to dissolve 5.57 g of BaCO3? 2HNO3(aq) +
Fantastic news! We've Found the answer you've been seeking!
Question:
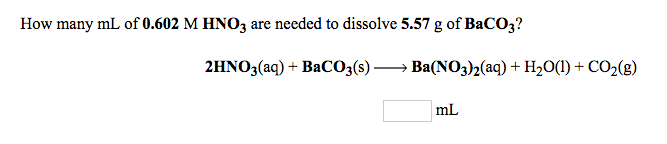
How many mL of 0.602 M HNO3 are needed to dissolve 5.57 g of BaCO3? 2HNO3(aq) + BaCO3(s) Ba(NO3)2(aq) + H2O(l) + CO2(g) mL
Related Book For 

Posted Date:




