Question: 1. Assume that (x,y,z,t) = (x-z) t i + (y +z)tj +(x+y) k. Determine the velocity and acceleration at (1,4,2,1).
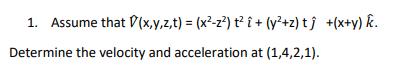
1. Assume that (x,y,z,t) = (x-z) t i + (y +z)tj +(x+y) k. Determine the velocity and acceleration at (1,4,2,1).
Step by Step Solution
3.50 Rating (157 Votes )
There are 3 Steps involved in it
To find the velocity and acceleration at 1421 we need to evaluate the partial derivatives of x y z a... View full answer

Get step-by-step solutions from verified subject matter experts


