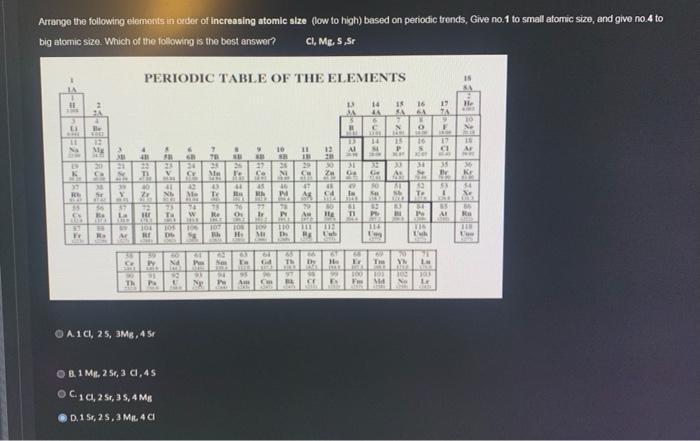
Arrange the following elements in order of increasing atomic size (low to high) based on periodic...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
Arrange the following elements in order of increasing atomic size (low to high) based on periodic trends, Give no.1 to small atomic size, and give no.4 to big atomic size. Which of the following is the best answer? CI, Mg, S,Sr PERIODIC TABLE OF THE ELEMENTS 11 30 -3=2²0Det & Fr 2 4 Be VORE 32 Mg PUS 20 23 G 54 TI 45 39 38 St 95 Ba ANTA - Rə A 22 102 $7 La 40 M 72 HE ALE TERD 38 59 Ce Py 2003 90 Th PA 16 A1CI, 25, 3MB, 4 Sr -=95PATE B.1 Mg, 2 5r, 3 cl, 45 C10, 2 Sr, 35, 4 Mg D.1 Sr, 25, 3 Mg. 4 Cl FIL No 60 PN 6 1001 92 129 24 35 16 42 Me 2014 M THE B 192 101 105 106 107 108 109 BE Db Bh H₂ B 7 70 11 24 Ma Xe 16 43 Te Ra Np 78 Re FM. 11 THE 16 LIBE Pas Sex Da Pr 10 6 NO 27 Se 76 23 MIN 45 RA P MBER ez zirci24 19 PD 88 95 Am On 26 le Pr Ni an 110 D 65 Th 303 BL 25+2= Dy 13 46 13 W VF 11 12 20 30 31 FISER 13 APARA là He IV MIC Ga 49 In 1144 61 TI il 100 150 RED BA F Md 18 SA 64 N AME *** 33 15 16 P 8 M 91 L O B3 IN THE SE ANDE 34 S AL 83 56 To en e I 116 Ish 20 Yh La him 103 JOLK No Le ject-01001 450 He KIME 10 *108 15 Ar NOT 36 Kr DER 54 Xe LORE 99 Ku ATE Eve Arrange the following elements in order of increasing atomic size (low to high) based on periodic trends, Give no.1 to small atomic size, and give no.4 to big atomic size. Which of the following is the best answer? CI, Mg, S,Sr PERIODIC TABLE OF THE ELEMENTS 11 30 -3=2²0Det & Fr 2 4 Be VORE 32 Mg PUS 20 23 G 54 TI 45 39 38 St 95 Ba ANTA - Rə A 22 102 $7 La 40 M 72 HE ALE TERD 38 59 Ce Py 2003 90 Th PA 16 A1CI, 25, 3MB, 4 Sr -=95PATE B.1 Mg, 2 5r, 3 cl, 45 C10, 2 Sr, 35, 4 Mg D.1 Sr, 25, 3 Mg. 4 Cl FIL No 60 PN 6 1001 92 129 24 35 16 42 Me 2014 M THE B 192 101 105 106 107 108 109 BE Db Bh H₂ B 7 70 11 24 Ma Xe 16 43 Te Ra Np 78 Re FM. 11 THE 16 LIBE Pas Sex Da Pr 10 6 NO 27 Se 76 23 MIN 45 RA P MBER ez zirci24 19 PD 88 95 Am On 26 le Pr Ni an 110 D 65 Th 303 BL 25+2= Dy 13 46 13 W VF 11 12 20 30 31 FISER 13 APARA là He IV MIC Ga 49 In 1144 61 TI il 100 150 RED BA F Md 18 SA 64 N AME *** 33 15 16 P 8 M 91 L O B3 IN THE SE ANDE 34 S AL 83 56 To en e I 116 Ish 20 Yh La him 103 JOLK No Le ject-01001 450 He KIME 10 *108 15 Ar NOT 36 Kr DER 54 Xe LORE 99 Ku ATE Eve
Expert Answer:
Answer rating: 100% (QA)
Atomic size atomic radius decreases from left to right in a period in the periodic ... View the full answer

Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
How many moles of Al can be produced from 10.87 g of Ag? Al(NO3) 3(s) + 3Ag ( Al + 3AgNO3
-
How many moles of HCl can be produced from 0.226 g of SOCl2? SOCl2() + H2O() ( SO2(g) + 2HCl(g)
-
How many kilograms of iron can be produced from 2.00 kg of hydrogen, H2, when you reduce iron (III) oxide?
-
e) Based on your investigation of the data set create two well-formatted tables that illustrate some important features of the data. Your tables should have informative labels and a readable number...
-
Norfolk, Virginia, a major seaport on the East Coast, has a ship coal-loading facility. Coal trucks filled with coal arrive at the port facility at the mean rate of 149 per day (Poisson distributed)....
-
The point of focus is the notion of "honor" in Europe during the 17th & 18th centuries. I would like some discussion of this element of European society especially as you read about Europes conquest...
-
Consider the following cash flow profile and assume MARR is 10 percent/year and the finance rate is 4 percent/ year. a. Determine the MIRR for this project. b. Is this project economically...
-
Francisco Company uses the columnar cash journals illustrated in the textbook. In April, the following selected cash transactions occurred. 1. Made a refund to a customer as an allowance for damaged...
-
1. You invest $2000 in an account that pays simple interest of 7% for 10 years. The amount of money you'll have at the end of 10 years is: 2. Use the compound interest formula to determine the...
-
You are a CPA student working in the accounting department of Reynolds Liquor Distributors Ltd. (RLDL) for the year ended December 31, 2019. Your role requires you to prepare and analyze the annual...
-
Global Wood Transport Co. (GWT) is considering investing on a new machinery aiming to improve the efficiency of its production process and the quality of its products. The marketing department claims...
-
What is Costco's top 3 threats for the company?
-
Leonardo, who is married but files separately, earns $170,000 of taxable income. He also has $14,750 in city of Tulsa bonds. His wife, Theresa, earns $82,000 of taxable income. If Leonardo and his...
-
Smith Co. paid $100,000 to acquire land, building, and equipment. At the time of the acquisition, appraisal values for the individual assets were determined as: land, $30,000; building, $60,000; and...
-
A firm has total assets worth $2 million and its leverage ratio is 1.25. The firm's EBIT is $450,000 and interest expense is $50,000. The firm's tax rate is 40%. What is the firm's return on equity?...
-
Caterpillar (CAT) announced on Dec. 12, 2012 that it would pay a last quarter dividend of $0.52 on Dec. 31, 2012. The date of record was Dec. 24, 2012. Katy bought 300 shares on Nov. 15, John sold...
-
A steel beam used in a bridge is constructed in the summer at a temperature of 35 C. The temperature decreases to -15C in the winter. As a Civil Engineer, you have been asked to design the expansion...
-
Heineken N.V., a global brewer based in the Netherlands, reports the following balance sheet accounts for the year ended December 31, 2016 (euros in millions). Prepare the balance sheet for this...
-
Which of the following amino acids has a polar side chain? NH2 HANCH-CH-CH,CH-CH NH2 CHHH CH CH3
-
If 1-mol samples of urea, a nonelectrolyte, sodium chloride, and calcium chloride are each dissolved in equal volumes of water in separate containers:
-
Selenium tetrafluoride, SeF4, is a colorless liquid. It has a vapor pressure of 757 mmHg at 105C and 522 mmHg at 95C. What is the heat of vaporization of selenium tetrafluoride?
-
Because of the negative incentive effect that taxes have on goods with elastic supply, in the late 1980s Margaret Thatcher (then prime minister of Great Britain) changed the property tax to a poll...
-
Many of the buildings in Paris have Mansard roofs, such as those shown in the photograph on page 147. a. What property tax structure would bring this about? b. Could you imagine a change in the...
-
On September 29, 2008, the stock market fell almost 7%. How can we reconcile these kinds of huge losses in the stock market with the efficient market hypothesis?

Study smarter with the SolutionInn App


