Consider the following reaction: HO (1) H(g) + O (g) AH = +285.8 kJ mol-...
Fantastic news! We've Found the answer you've been seeking!
Question:
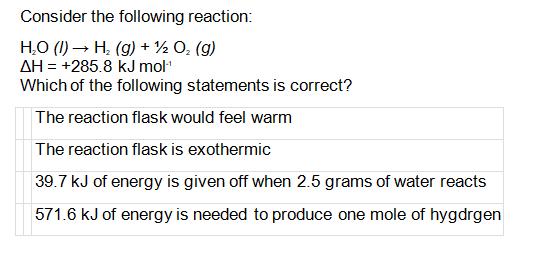
Transcribed Image Text:
Consider the following reaction: H₂O (1)→ H₂(g) + ½ O₂ (g) AH = +285.8 kJ mol-¹ Which of the following statements is correct? The reaction flask would feel warm The reaction flask is exothermic 39.7 kJ of energy is given off when 2.5 grams of water reacts 571.6 kJ of energy is needed to produce one mole of hygdrgen Consider the following reaction: H₂O (1)→ H₂(g) + ½ O₂ (g) AH = +285.8 kJ mol-¹ Which of the following statements is correct? The reaction flask would feel warm The reaction flask is exothermic 39.7 kJ of energy is given off when 2.5 grams of water reacts 571.6 kJ of energy is needed to produce one mole of hygdrgen
Expert Answer:
Answer rating: 100% (QA)
The reaction is endothermic because the enthalpy change is positive ... View the full answer

Related Book For 

Ethics in Accounting A Decision Making Approach
ISBN: 978-1118928332
1st edition
Authors: Gordon Klein
Posted Date:
Students also viewed these chemistry questions
-
What do you think about the idea of codes of conduct? Give three reasons why an organization ought to have a code of conduct, and give three reasons why an organization should not have a code of...
-
What do you think about Wholesomes idea for opening a new distribution channel?
-
You have a very outgoing personality and are extremely friendly with a staff member named Adelana. Adelana reports directly to you in the company's Billing Department. You are a CPA, but Adelana is...
-
A particle of mass m moves along the x-axis under a force F = ??m?2x. The amplitude is b. Show that the period is given by 4. T= dx, (11.83) where (11.84) Y =1+ 2c2 (2-2).
-
Estimate the enthalpy change for this reaction. Start by drawing the Lewis electron dot diagrams for each substance. 4NH3 + 3O2 ( 2N2 + 6H2O
-
Prepare a indirect cash flow statement Additional Information Sold available for sale securities (not cash equivalents costing $87,750 for $90,000 Equipment costing $18,750 with a book value of $3750...
-
The plaintiff, Thelma Agnes Smith, lived with the defendant out of wedlock for several years. When the relationship ended, she sued the defendant, seeking to enforce two written agreements with him...
-
Trendy Toes, Co., produces sports socks. The company has fixed costs of $95,000 and variable costs of $0.95 per package. Each package sells for $1.90. Requirements 1. Compute the contribution margin...
-
If your car gets 25 miles per gallon, how much does it cost to drive 420 miles when gasoline costs $2.50 per gallon? The cost is $ (Simplify your answer. Round to the nearest cent as needed.)
-
Lido Company's standard and actual costs per unit for the most recent period, during which 400 units were actually produced, are given below: Standard Actual Materials: Standard: 2 foot at $1.50 per...
-
(1) The ratio of sex between men and women is known to be 1:1. What is the probability that a family with six children now has three sons and three daughters? (2) Calculate probability for x=5 by...
-
The market and Stock J have the following probability distributions: Probability r M r J 0.3 15 % 21 % 0.4 9 3 0.3 19 11 Calculate the expected rate of return for the market. Round your answer to two...
-
A small object begins a free-fall from a height of 22.0 m. After 1.10 s, a second small object is launched vertically upward from the ground with an initial velocity of 32.0 m/s. At what height h...
-
Bob exchanged a building for another building on April 3, 2023. The adjusted basis of his building was $320,000. He also gave $50,000 cash. He received a building with a fair market value of...
-
Given the following piecewise function: f(x) 3 Find the value of ' (-27) + ' (7) 2 = { 2 sin(x), if x 0 3 cos(x), if x > 0
-
Assume that your cousin holds just one stock, Eastman Chemical Bonding (ECB), which he thinks has very little risk. You agree that the stock is relatively safe, but you want to demonstrate that his...
-
5. A US bank is subject to two restrictions: a required reserve ratio of 20% and the limit on the deposit rate of 6% per year. Suppose that a non-US branch of the bank does not have to put any...
-
31. What is the income that can be received over 15 years from $500,000 earning 6% annually? 32. What is the semiannual payment required to retire $50,000 in debt over 5 years at 8% compounded...
-
As a student, you have the opportunity to work in the Volunteer Income Tax Assistance Program, commonly known as VITA. VITA volunteers principally prepare tax returns for indigent taxpayers, free of...
-
Section 5061(a) of California's Business and Professions Code states that "a person engaged in the practice of public accountancy shall not ... pay a fee or commission to obtain a client." You are a...
-
Before issuance, most corporate and governmental bonds receive a credit rating that usually ranges from a high of triple-A down to B-. Nearly all bond ratings are issued by one of three credit rating...
-
What might be problematic about these responses to interview questions? How might the answers be improved? a. Q: Tell me about yourself. A: Im really easy-going and casual. b. Q: I noticed that you...
-
Revise the follow-up message below to be more professional and effective, based on the principles in this chapter. December 2, 2021 Ms. Charlotte LeClaire Pebble Creek Publishing Inc. New York, NY...
-
Write a follow-up email message or letter after an office visit or plant trip. Thank your hosts for their hospitality; relate your strong points to things you learned about the company during the...

Study smarter with the SolutionInn App


