Identify (circle) the oxidizing agent in each of the following redox reactions: Zn (s) + F,...
Fantastic news! We've Found the answer you've been seeking!
Question:
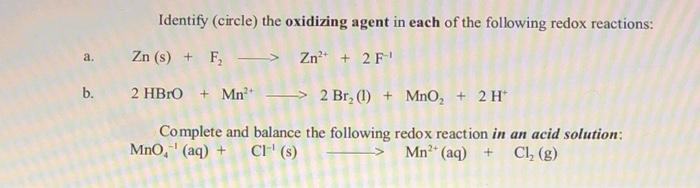
Transcribed Image Text:
Identify (circle) the oxidizing agent in each of the following redox reactions: Zn (s) + F, > Zn* + 2 F a. b. 2 HBRO + Mn > 2 Br, (1) + MnO, + 2 H* Complete and balance the following redox reaction in an acid solution: MnO, (aq) + CI" (s) > Mn (aq) + Cl, (g) Identify (circle) the oxidizing agent in each of the following redox reactions: Zn (s) + F, > Zn* + 2 F a. b. 2 HBRO + Mn > 2 Br, (1) + MnO, + 2 H* Complete and balance the following redox reaction in an acid solution: MnO, (aq) + CI" (s) > Mn (aq) + Cl, (g)
Expert Answer:
Answer rating: 100% (QA)
Oxidising agent is that reactant which helps oxidation by accepting electrons and itself get reduced ... View the full answer

Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
Circle the isoprene units in each of the following compounds. (a) (b) (c) Menthol HO Grandisol
-
In each of the following reactions, an amine or a lithium amide derivative reacts with an aryl halide. Give the structure of the expected product, and specify the mechanism by which it is formed.
-
Identify the ethical dilemmas in each of the following situations. (A situation might involve more than one dilemma.) a. Due to the breakup with a client, an advertising agency finds itself working...
-
The monthly earnings of financial analysts are normally distributed with a mean of $5,700. If only 6.68 % of the financial analysts have a monthly income of more than $6,140, what is the value of the...
-
The Loyd Company had 150 units of product Omega on hand at December 1, Year 1 costing $ 400 each. Purchases of product Omega during December were as follows: Sales during December were 500 units on...
-
Given the function f(x) = ax + b, a 0, find the values of a and b for which f -1 (x) = f(x).
-
Distinguish between logical and physical models. a. Which one comes first? b. Where do conceptual models fit?
-
Prepare journal entries to record the following merchandising transactions of Cabela's, which uses the perpetual inventory system and the gross method. July 1 Purchased merchandise from Boden Company...
-
What mechanisms can be implemented to ensure accountability and mitigate the risks associated with delegation, such as delegation without proper oversight or the delegation of inappropriate tasks ?
-
You are creating a customer database for the Lehigh Valley IronPigs minor league baseball team. Draw a project network given the information below. Complete the forward and backward pass, compute...
-
A simple pendulum consisting of a 10 m long string and a 0.20 kg bob is pulied to the side so that the string makes an angle of 20 with the vertical If the grevitational potential energy of the...
-
The following AVL tree is not balanced. Redraw the tree after rebalancing. You do not have to list the balance factors. 5 15) 30 20 25 50
-
A property forecasted to produce $44,555 of gross income, and which sells for $412,450 is said to have sold at a GIM of?
-
A channel is constructed of unfinished concrete. It is 10 ft wide, has a slope of 0.005 and carries a flow of 120 cu.ft/s. The flow encounters an obstruction that rises 0.5 ft above the channel...
-
EXCEL EXERCISES 35 15.) The financial records of the Colonial Restaurant reveal the following figures for the year ending December 31, 20XX: Depreciation, $25,000 Food sales, $375,000 Cost of...
-
CNNs capital structure is made of 50% ordinary shares; 30% preference share and 20% debt, cost of debt is 9 per cent. The risk-free rate is 3%, beta of CNN is 1.20 and the market risk premium is 12%....
-
Do you agree or disagree with the statement that Lancewood for diary products can use the ABC inventory analysis for managing independent finished goods?
-
Determine whether the lines are parallel, perpendicular, or neither. 2x + 3y = -12, 2y - 3x = 8
-
(a) Two isomeric S"2 products are possible when sodium thiosulfate is allowed to react with one equivalent of methyl iodide in methanol solution. Give the structures of the two oroducts. ...
-
When p-aminophenol reacts with one molar equivalent of acetic anhydride, a compound acetaminophen (A, C8H9NO2) is formed that dissolves in dilute NaOH. When A is treated with one equivalent of NaOH...
-
Consider two 2p orbitals, one on each of two different atoms, oriented side-to-side, as in Figure Pl.46. Imagine bringing these nuclei together so that overlap occurs as shown in the figure. This...
-
There are 24,627 species of fish on Earth. Decide whether the statement makes sense (or is clearly true) or does not make sense (or is clearly false). Explain clearly.
-
For a flight on a small plane, the pilot asks passengers what they weigh. Identify at least one likely source of random errors and also identify at least one likely source of systematic errors.
-
An American Airlines agent tells you that you must pay a surcharge because your checked bag weighs 23.018 kg, which exceeds the limit of 23 kg, and that theres no doubt that the scale is correct...

Study smarter with the SolutionInn App


