If 23.2 ml of NaOH solution is required to neutralize 0.123 g of KHP (molar mass...
Fantastic news! We've Found the answer you've been seeking!
Question:

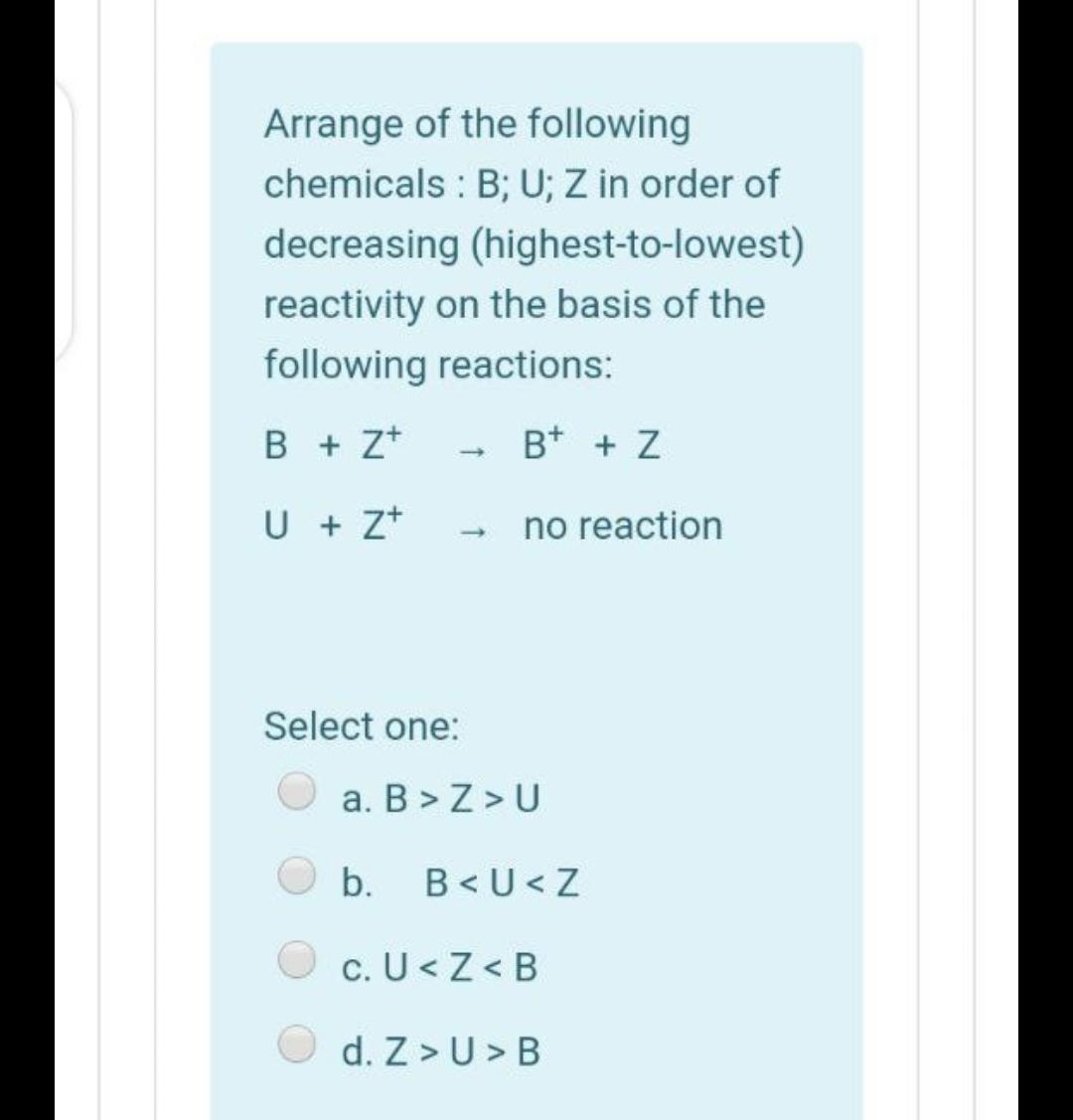
Transcribed Image Text:
If 23.2 ml of NaOH solution is required to neutralize 0.123 g of KHP (molar mass 204.2 g/mol), then the molarity of NaOH is: Select one: a. 0.026 b. 2.60 c. 0.922 d. 0.014 Arrange of the following chemicals : B; U; Z in order of decreasing (highest-to-lowest) reactivity on the basis of the following reactions: B + z* B+ + Z U + z+ no reaction Select one: a. B > Z > U b. B<U< Z c. U<Z< B d. Z >U>B If 23.2 ml of NaOH solution is required to neutralize 0.123 g of KHP (molar mass 204.2 g/mol), then the molarity of NaOH is: Select one: a. 0.026 b. 2.60 c. 0.922 d. 0.014 Arrange of the following chemicals : B; U; Z in order of decreasing (highest-to-lowest) reactivity on the basis of the following reactions: B + z* B+ + Z U + z+ no reaction Select one: a. B > Z > U b. B<U< Z c. U<Z< B d. Z >U>B
Expert Answer:

Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
What volume of 0.0521 M Ba(OH) 2 is required to neutralize exactly 14.20 mL of 0.141 M H 3 PO 4 ? Phosphoric acid contains three acidic hydrogens.
-
Arrange the following compounds in order of decreasing SN2 reactivity toward sodium ethoxide: CH3 CH3 CH3CH2CHB CH CHCH2Br CH,CH2CH2CH2Br
-
Arrange the following alkyl bromides in order of decreasing reactivity in an SN1 reaction: isopropyl bromide, propyl bromide, tert-butyl bromide, methyl bromide.
-
A company writes a check to replenish a $100 petty cash fund when the fund contains receipts of $94 and $4 in cash. In recording the check, the company should: (a) debit Cash Over and Short for $2....
-
For the 18-hole courses in file golfsrs.dat, plot the weekend 18-hole greens fee vs. the backtee yardage. Estimate the regression parameters for predicting weekend greens fees from backtee yardage....
-
What is the purpose of the balance sheet? Briefly identify and describe the major types of assets and the claims of creditors and owners shown on the typical balance sheet.
-
This case stems from a rocky relationship between two Texans. Lawrence Shipley was the president of Shipley Do-Nut Flour & Supply Co., and Andrea Vasquez was a substitute school teacher in the...
-
Rapid Auto has over 200 auto-maintenance service outlets nationwide. It provides primarily two lines of service: oil changes and brake repair. Oil change-related services represent 65% of its sales...
-
Nick went out for dinner with a few friends. The restaurant bill should have been $175, but instead, the waitress brings the bill for $160. Find the absolute error and relative percentage error for...
-
Lee, Danny and Sue were childhood friends. They loved horses and dreamed of one day owning a horse who won the huge prize money in the famous annual Melbourne Cup horse race. To fulfill this dream,...
-
The Widget Compan y is a small company with only a few employees. Its line of business is to purchase several items from a line of widgets and resale them to other companies. The Company owns one...
-
Durable Plastics Company had the following total assets, liabilities, and equity as of December 31. Total Assets $ 440,000 Total Liabilities 133,000 Total Equity 307,000 What is the company's debt...
-
You are the sole contributor to a philanthropic foundation. You must specify whether investment gains should be expendable or non-dependable. Present the key argument in favor and against permitting...
-
A parallel plate capacitor with an air gap is connected to a 6V battery. The charged capacitor stores energy of 72nJ. Without disconnecting the capacitor from the battery, a dielectric material is...
-
What are the different ways that cells can accomplish local and long distance signaling?
-
A friend of yours is just starting a delivery service and has purchased a truck for $20,000. The friend expects the truck to last 5 years and to have no resale value at that point. The friend's...
-
Jill has just won the lottery. She will receive semi-annual payments of $50,000 commencing in 6 months time and growing at 1.4% each half-year, forever. If the interest rate is 12.9% per annum...
-
For the next several days, take notes on your listening performance during at least a half-dozen situations in class, during social activities, and at work, if applicable. Referring to the traits of...
-
Element X forms both a dichloride (XCl2) and a tetra-chloride (XCl4). Treatment of 10.00 g of XCl2 with excess chlorine forms 12.55 g of XCl4. Calculate the atomic weight (mass) of X and identify X.
-
The density of a pure gaseous compound was measured at 0.00 o C as a function of pressure to give the following results: Density (g/ L) . Pressure (atm) 0.17893...
-
The designations 1A through 8A used for certain families of the periodic table are helpful for predicting the charges on ions in binary ionic compounds. In these compounds, the metals generally take...
-
Goods are products a business sells like a. haircuts. b. conveyer belts. c. car insurance. d. health care.
-
Which of the following are the factors of production? a. Labor, natural resources, capital, entrepreneurs, technology, and intellectual property b. Labor, capital, entrepreneurs, motivation, and good...
-
Which of the following is a current sociocultural trend? a. A decrease in the overall U.S. population b. An increase in the population of Americans ages 30 to 45 years old c. A decrease in the U.S....

Study smarter with the SolutionInn App


