Given that Cv = ( at constant volume: dU = CydT and f dU = AU...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
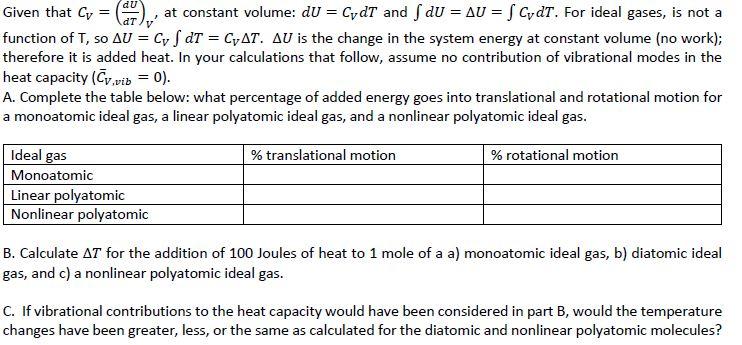
Given that Cv = ( at constant volume: dU = CydT and f dU = AU = f CydT. For ideal gases, is not a function of T, so AU = Cyf dT = CAT. AU is the change in the system energy at constant volume (no work); therefore it is added heat. In your calculations that follow, assume no contribution of vibrational modes in the heat capacity (Cv.vib = 0). A. Complete the table below: what percentage of added energy goes into translational and rotational motion for a monoatomic ideal gas, a linear polyatomic ideal gas, and a nonlinear polyatomic ideal gas. Ideal gas Monoatomic Linear polyatomic Nonlinear polyatomic % translational motion % rotational motion B. Calculate AT for the addition of 100 Joules of heat to 1 mole of a a) monoatomic ideal gas, b) diatomic ideal gas, and c) a nonlinear polyatomic ideal gas. C. If vibrational contributions to the heat capacity would have been considered in part B, would the temperature changes have been greater, less, or the same as calculated for the diatomic and nonlinear polyatomic molecules? Given that Cv = ( at constant volume: dU = CydT and f dU = AU = f CydT. For ideal gases, is not a function of T, so AU = Cyf dT = CAT. AU is the change in the system energy at constant volume (no work); therefore it is added heat. In your calculations that follow, assume no contribution of vibrational modes in the heat capacity (Cv.vib = 0). A. Complete the table below: what percentage of added energy goes into translational and rotational motion for a monoatomic ideal gas, a linear polyatomic ideal gas, and a nonlinear polyatomic ideal gas. Ideal gas Monoatomic Linear polyatomic Nonlinear polyatomic % translational motion % rotational motion B. Calculate AT for the addition of 100 Joules of heat to 1 mole of a a) monoatomic ideal gas, b) diatomic ideal gas, and c) a nonlinear polyatomic ideal gas. C. If vibrational contributions to the heat capacity would have been considered in part B, would the temperature changes have been greater, less, or the same as calculated for the diatomic and nonlinear polyatomic molecules?
Expert Answer:
Answer rating: 100% (QA)
A Complete the table below what percentage of added energy goes into transl ational and rot ational motion for a mon oat omic ideal gas a linear poly ... View the full answer

Related Book For 

Posted Date:
Students also viewed these accounting questions
-
2. Suppose money demand function is given by M-500*(0.25-r), r is real interest rate, money supply M' is 100 (billion dollars) and the required reserve ratio is 20%. a) Calculate equilibrium interest...
-
The molar heat capacity of a diatomic molecule is 29.1 J/K mol. Assuming the atmosphere contains only nitrogen gas and there is no heat loss, calculate the total heat intake (in kilojoules) if the...
-
The vibrational contribution to the molar heat capacity of a gas of nonlinear molecules is given in statistical mechanics by the formula i=1 " style="" class="fr-fic fr-dib"> where u i =...
-
Consider examples 9.2 and 9.3. Assume that social damage is quadratic as in the examples. Following section 9.2.3, assume that the regulator can only set a uniform tax. Determine the formula for the...
-
Littlerock Surveying showed the following partial unadjusted results at May 31, 2014, its month-end: Part 1 Required a. Assuming Littlerock estimates bad debts to be 2.5% of sales, prepare the...
-
Bernie and Phils Great American Surplus store placed an ad in the Sunday Times stating, Next Saturday at 8:00 A.M. sharp, 3 brand new mink coats worth $5,000 each will be sold for $500 each! First...
-
In a contract dispute between a US company and a Canadian company, the contract itself referred to provisions of the Uniform Commercial Code. Do these references alone preempt the contract from being...
-
Information related to Harwick Co. is presented below. 1. On April 5, purchased merchandise from Botham Company for $23,000, terms 2/10, net/30, FOB shipping point. 2. On April 6, paid freight costs...
-
A trebuchet is a mighty siege engine capable of flinging 90-kg projectiles a distance of 300 meters. Assuming a perfect launch angle of 45, calculate the speed with which one of these projectiles...
-
Using Bayes decision rule, consider the decision analysis problem having the following payoff table (in units of thousands of dollars): (a) Which alternative should be chosen? What is the resulting...
-
Which of the following statements is CORRECT? The value of operations of a stock is the present value of all expected future free cash flows, discount at the free cash flow growth rate. The free cash...
-
What is the frequency of light with a wavelength of 756nm?
-
Q1. Identify and explain the major factors motivating firms' international location decision strategies (e.g., manufacturing internationally) in many countries such as the UAE. Q2. Identify and...
-
A chunk of nickel weighing 19.0 grams and originally at 98.12 C is dropped into an insulated cup containing 83.6 grams of water at 21.62 C. Assuming that all of the heat is transferred to the water,...
-
42. [II] Three point-charges are placed at the corners of an isosceles triangle. At the left and right, end points of the base are +1.0 C and +1.0 C, respectively, and at the vertex +3.0 C. The base...
-
Identify two initiatives that Trinity Health should pursue next to leverage its existing IT capability that would increase the quality of care or reduce cost. Describe why you selected the...
-
Examples of organizations using substitute items since 2020 (COVID)?
-
B.) What is the approximate concentration of free Zn 2+ ion at equilibrium when 1.0010 -2 mol zinc nitrate is added to 1.00 L of a solution that is 1.080 M in OH - . For [Zn(OH) 4 ] 2- , K f = 4.610...
-
Aqueous copper(II) sulfate solution is blue in color. When aqueous potassium fluoride is added, a green precipitate is formed. When aqueous potassium chloride is added instead, a bright-green...
-
Assuming ideal behavior, calculate the density of gaseous HF at its normal boiling point (19.5C). The experimentally measured density under the same conditions is 3.10 g/L. Account for the...
-
Calculate the molecular mass or formula mass (in amu) of each of the following substances: (a) CH4, (b) NO2, (c) SO3, (d) C6H6, (e) NaI, (f) K2SO4, (g) Ca3 (PO4)2.
-
Rewrite the Gross-Pitaevskii equation and the mean field energy, see equations (11.2.21) and (11.2.23), for an isotropic harmonic oscillator trap with frequency \(\omega_{0}\) in a dimensionless form...
-
The energy levels of an imperfect Fermi gas in the presence of an external magnetic field \(\boldsymbol{H}\), to the first order in \(a\), may be written as...
-
Solve the Gross-Pitaevskii equation (11.2.23) in a harmonic trap for the case when the scattering length \(a\) is zero. Show that this reproduces the properties of the ground state of the...

Study smarter with the SolutionInn App


