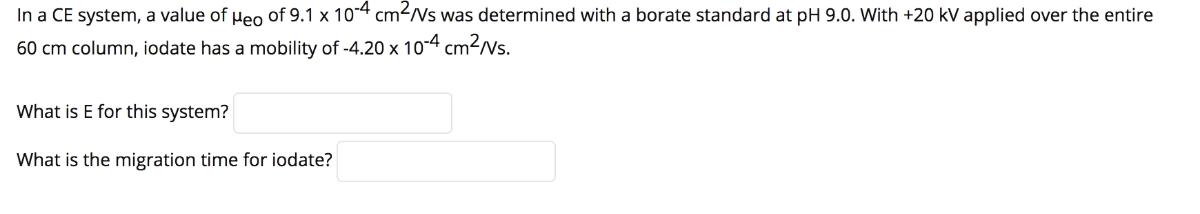
In a CE system, a value of ueo of 9.1 x 104 cm-/Vs was determined with...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
In a CE system, a value of ueo of 9.1 x 104 cm-/Vs was determined with a borate standard at pH 9.0. With +20 kV applied over the entire 60 cm column, iodate has a mobility of -4.20 x 10-4 cm?/Vs. What is E for this system? What is the migration time for iodate? In a CE system, a value of ueo of 9.1 x 104 cm-/Vs was determined with a borate standard at pH 9.0. With +20 kV applied over the entire 60 cm column, iodate has a mobility of -4.20 x 10-4 cm?/Vs. What is E for this system? What is the migration time for iodate?
Expert Answer:
Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
4 cubed + 10 X 20 + 8 squared 23
-
A saturated solution of copper(II) iodate in pure water has a copper-ion concentration of 2.7 10-3 M. a. What is the molar solubility of copper iodate in a 0.35 M potassium iodate solution? b. What...
-
A saturated solution of lead iodate in pure water has an iodate-ion concentration of 8.0 10-5 M. a. What is the molar solubility of lead iodate in a 0.15 M lead nitrate solution at the same...
-
Marty owns an auto body repair shop. The business has no real value without him and the building is pretty much a mess after being full of paint fumes for all those years but the land is worth...
-
The female head of the physical therapy department at a medical clinic encountered problems with a male chiropractor who also worked at the clinic. The chiropractor regularly put his arm around and...
-
Suppose that the range of g lies in the domain of so that the composite g is defined. If and g are one-to-one, can anything be said about g? Give reasons for your answer.
-
For each of the following situations, calculate the degrees of freedom \((d f)\), identify the critical values (assume \(\mathbf{a}=.05\) [two-tailed]), calculate the standard error of the mean \(s\)...
-
At the end of its first year of operations, the Leo Company lists the following accounts and ending account balances related to stock transactions and dividends: During the first year the following...
-
Luccasen Inc., has a market beta of 1.25. The risk-free interest rate is 3%, and the market risk premium is 8%. Given this information, what is the expected return for Luccasen Inc.?
-
The cash book (bank columns) of Ella for the month of May 207 were as follows: Ellas bank statement for May 207 showed the following: The following errors were discovered: 1. Ella had overcast the...
-
I am supposed to do this but I have no idea what to do. Implement deadlock detection: Assume you have a database management system that implements a locking mechanism. You are running a deadlock...
-
1) Describe your company's stock repurchase policy over the last 3 years . 2) Describe the stock repurchase policy of your competitor(s) over the last 3 years . 3) Compare the stock repurchase policy...
-
[Acua] Consider the following array: 8, 9, 17, 4, 3, 20, 25, 5 Show a trace of execution for top-down mergesort using the method shown in lecture (where both sides are updated at once). Illustrate...
-
Give a short portrayal of the three sections that make up the Global Situating System (GPS), and their fundamental programming, equipment and data parts. [5 marks] ? [5 marks] (c) What information is...
-
The following transactions occurred in the accounts of UTOPOLO SC on June 2022 June1 Started in business with Bank Account of TZS20,000,000 and stock valued at TZS 300,000. GSM LTD lent UTOPOLO...
-
Describe the four access regimes from public to private that may be applied to Java fields and methods. Why are they useful? [4 marks] (b) When you extend a class, the constructor for your new class...
-
Problem 24: A ship 650 ft long is to operate at a speed of 22 mph in ocean water whose kinematic viscosity is 0.00001261 ft/s. What should be the kinematic viscosity of a fluid used with an 11-ft...
-
Briefly describe the following types of group life insurance plans: a. Group term life insurance b. Group accidental death and dismemberment insurance (AD&D) c. Group universal life insurance d....
-
If 20.0 mL of a 0.10 M NaOH solution is added to a 30.0- mL sample of a 0.10 M weak acid, HA, what is the pH of the resulting solution? (K a = 1.8 10 5 for HA) a. 2.87 b. 2.74 c. 4.74 d. 5.05 e. 8.73
-
Phosphorus pentachloride, PCl5, decomposes when heated. If the initial concentration of PCl5 is 1.00 mol/L, what is the equilibrium composition of the gaseous mixture at 160oC? The equilibrium...
-
Starting with Charless law (stated as an equation), obtain an equation for the final volume of a gas from its initial volume when the temperature is changed at constant pressure.
-
Has the U.S. economy experienced inflation or deflation during recent recessions? Explain.
-
How do you think recessions influence elections?
-
Inflation is soaring and employment is beginning to show sustained improvement. The unemployment rate is 6 percent, compared to its 3.5 percent rate prepandemic. The CPI grew 5.4 percent in June, and...

Study smarter with the SolutionInn App



