In a HCI molecule, the energy gap between the two vibrational levels is eV. (Upto two...
Fantastic news! We've Found the answer you've been seeking!
Question:
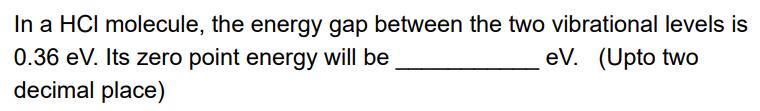
Transcribed Image Text:
In a HCI molecule, the energy gap between the two vibrational levels is eV. (Upto two 0.36 eV. Its zero point energy will be decimal place) In a HCI molecule, the energy gap between the two vibrational levels is eV. (Upto two 0.36 eV. Its zero point energy will be decimal place)
Expert Answer:
Related Book For 

Posted Date:
Students also viewed these physics questions
-
The three vibrational frequencies in H 2 O (1595, 3657, and 3756 cm 1 ) are all much larger than the corresponding frequencies in D 2 O (1178, 2671, and 2788 cm 1 ). This follows from the fact that...
-
The following additional information is available for the Dr. Ivan and Irene Incisor family from Chapters 1-5. Ivan's grandfather died and left a portfolio of municipal bonds. In 2012, they pay Ivan...
-
The energy gap between valence and conduction bands in germanium is 0.72eV. What range of wavelengths can a photon have to excite an electron from the top of the valence band into the conduction hand?
-
In Exercises 3542, describe and sketch the surface given by the function. f(x, y) = Jxy, 0, x 0, y 0 x <0 or y < 0
-
Inventory that has a market value below the historical cost should be written down in order to recognize a loss.
-
a. Use the distance formula to find the distance between (4a, a) and (3a, 2a). Hence find the distance between the following pairs of points: b. (4, 1) and (3, 2) c. (12, 3) and (9, 6) d. (20, 5) and...
-
Suppose that a one-way analysis of variance involves four treatments but that a different number of observations (e.g., $n_{i}$ ) has been taken under each treatment. Assuming that $n_{1}=3, n_{2}=2,...
-
Compare the advantages and disadvantages of using spot checks/hidden video cameras in the workplace and pay-for-performance pay schemes as means to influence worker performance.
-
Question: The following activities occur at Greenwich Corporation, a company that contracts out a temporary workforce to retailers to meet seasonal demand:Required:Classify each of the activities...
-
A convenience store is considering changing its layout to encourage impulse buying. The triangular flow matrix below gives the measure of association between different product groups ( e. g., beer,...
-
using the combined formulas for the breakeven units (fixed costs contribution margin per unit) and breakeven revenues (breakeven units x selling price), calculate the present breakeven point in...
-
How are fixed and variable materials costs distinguished?
-
Suppose that we wish to compare the health status of two groups of people. What variable might we use to measure the status? What variables might we wish to control in order to draw the appropriate...
-
You are the newly appointed secretary of a primary school employing 20 teachers and having 300 pupils. The head teacher has asked you to design a system for ordering books and stationery and...
-
Analyze how social media could influence each stage of the Customer Decision Journey for a customer deciding where to go on vacation.
-
From the following trial balance of G. Still, draw up an income statement for the year ending 30 September 2013, and a statement of financial position as at that date. Inventory: 1 October 2012...
-
The borrowers First Mortgage Balance is $200,000. They are going to subordinate a HELOC that has a Current balance of $50,000 and a total Line amount of $75,000. The property appraised at $550,000....
-
The diameter of a sphere is 18 in. Find the largest volume of regular pyramid of altitude 15 in. that can be cut from the sphere if the pyramid is (a) square, (b) pentagonal, (c) hexagonal, and (d)...
-
DielsAlder cycloaddition of 1, 3-butadiene with acrylonitrile requires that the diene be in a cis (or cis-like) conformation: In fact, the diene exists primarily in a trans conformation, the cis...
-
In the neck of the flask depicted in the text, five red balls rest on five blue balls. Suppose the balls are tipped back into the flask, shaken, and the flask is re-inverted. Whats the probability...
-
Assume that 1-bromobutane and 1-chlorobutane form an ideal solution. At 273 K, P * chloro = 3790. Pa and P * bromo = 1394 Pa. When only a trace of liquid is present at 273 K, y chloro = 0.750. a....
-
An article on barrons.com observed that the U.S. dollar has been droppingand thats good news for the stock market and companies that get a large chunk of their sales from overseas. a. What does the...
-
In May 2023, the Bureau of Economic Analysis (BEA) issued a revised estimate of the change in real GDP during the first quarter of 2023. The BEAs news release describing the revisions noted that the...
-
During the second quarter of 2020, real inventories declined by $274 billion, by far the largest decline on record. During the third quarter of 2020, real GDP increased by 35 percent, by far the...

Study smarter with the SolutionInn App



