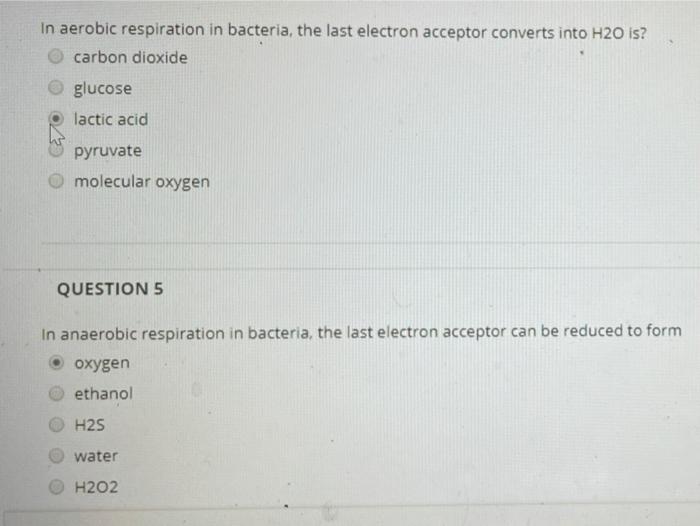
In aerobic respiration in bacteria, the last electron acceptor converts into H2O is? carbon dioxide glucose...
Fantastic news! We've Found the answer you've been seeking!
Question:

Transcribed Image Text:
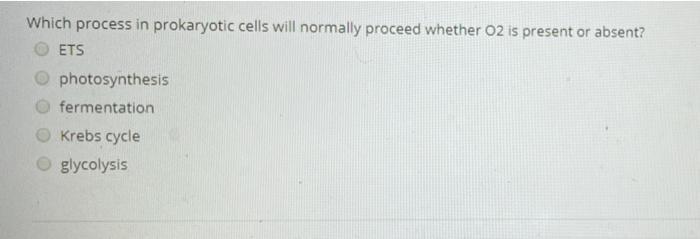
In aerobic respiration in bacteria, the last electron acceptor converts into H2O is? carbon dioxide glucose lactic acid pyruvate molecular oxygen QUESTION 5 In anaerobic respiration in bacteria, the last electron acceptor can be reduced to form oxygen ethanol H2S water H202 if there is no 02, then CAC: Fermentation ETS: Fermentation None Acetyl-CoA: Fermentation will not occur and the pyruvate from glycolysis is used by under anaerobic conditions Which process in prokaryotic cells will normally proceed whether O2 is present or absent? ETS photosynthesis fermentation Krebs cycle glycolysis In aerobic respiration in bacteria, the last electron acceptor converts into H2O is? carbon dioxide glucose lactic acid pyruvate molecular oxygen QUESTION 5 In anaerobic respiration in bacteria, the last electron acceptor can be reduced to form oxygen ethanol H2S water H202 if there is no 02, then CAC: Fermentation ETS: Fermentation None Acetyl-CoA: Fermentation will not occur and the pyruvate from glycolysis is used by under anaerobic conditions Which process in prokaryotic cells will normally proceed whether O2 is present or absent? ETS photosynthesis fermentation Krebs cycle glycolysis
Expert Answer:
Answer rating: 100% (QA)
In aerobic respiration in bacteria the last electron ac... View the full answer

Related Book For 

Posted Date:
Students also viewed these biology questions
-
Determine whether the force-and-couple system shown can be reduced to a single equivalent force R. If it can, determine R and the point where the line of action of R intersects the yz plane, if it...
-
Quinone can be reduced to hydroquinone with an excess of electrolytically generated Sn(II): The polarity of the working electrode is then reversed, and the excess Sn(II) is oxidized with Br2...
-
Determine whether the force-and-couple system shown can be reduced to a single equivalent force R. If it can, determine R and the point where the line of action of R intersects the yz-plane. If it...
-
When the freed slave saw the real physical object? A) He was perplexed by the object when he was told what they were B) He was dazzled by the light C) He didn't believe what he was told D) all adove...
-
Rachel Manny has prepared the following list of statements about the general ledger. 1. The general ledger contains all the asset and liability accounts, but no stockholders' equity accounts. 2. The...
-
The flash on a compact camera stores energy in a 120 F capacitor that is charged to 220 V. When the flash is fired, the capacitor is quickly discharged through a lightbulb with 5.0 of resistance. a....
-
Compute the point estimates b0 and b1. Exercises 610 refer to the following data set: x 25 13 16 19 29 19 16 30 y 40 20 33 30 50 37 34 37
-
Morrison and Amato have decided to form a partnership. They have agreed that Morrison is to invest $150,000 and that Amato is to invest $50,000. Morrison is to devote one-half time to the business...
-
If the nominal rate of return is 10.37% and the real rate of return is 6.68%, at what rate are costs changing?
-
Punnett Square 2 shows a Punnett square describing a cross between two individuals from the F 1 generation. Was Punnett square 2 a good predictor of offspring phenotypes? Support your answer by...
-
1. The number of components per hour turned on a lathe was measured for 40 hours 18 17 18 19 17 19 19 18 18 15 16 17 18 16 17 Compile a frequency table: compts /hr (x) 15 18 19 20 21 16 21 19 18 18...
-
A motorist traveling at 55 mph down a grade of 5% on a highway observes an accident involving an overturned truck ahead of him which completely blocked the road. If the motorist was able to stop his...
-
In this task, you are required to answer the questions by referring to the data table given below. The table shows 15 cases, college students, from a hypothetical study of student loan balances at...
-
A test of a driver's perception reaction time is being conducted on a special testing track with wet pavement and a driving speed of 55 mi/hr. When the driver is sober, a stop can be made just in...
-
Other Itemized Deductions (LO 5.10) During the 2023 tax year, Irma incurred the following expenses: Union dues Tax return preparation fee Brokerage fees for the purchase of stocks Uniform expenses...
-
How opening up to trade specifically affects a domestic monopoly. What is game theory and how even a single additional competitor can lead to a market outcome similar to perfect competition?
-
You decide you want to overclock the processors on your system. What is the first step in the process? O a. Boot Windows as Administrator, go to Control Panel, set the BIOS/UEFI flag, then restart O...
-
To help you become familiar with the accounting standards, this case is designed to take you to the FASBs Web site and have you access various publications. Access the FASBs Web site at...
-
Data on a single crystal of NiS047H20 give Xm= 6.00 X 10-8 m3 mol-l at 298 K. Determine the effective number of unpaired electrons in this compound and compare your result with the theoretical value.
-
Aromatic hydrocarbons and 12 form complexes from which charge transfer electronic transitions are observed the hydrocarbon acts as an electron donor and 12 as an electron acceptor the energies hvmax...
-
Demonstrate that the Planck distribution reduces to the Rayleigh-Jeans law at long wavelengths.
-
The presence of air in the brake system fluid causes the brake pedal to feel __________________.
-
Three main components of pst brake antiskid systems are __________________, __________________ and a __________________.
-
__________________ pressure bleeding is not an option for power brakes; the air trapped in the brake system would be forced into the main hydraulic system, which should operate without air in the...

Study smarter with the SolutionInn App


