In an attempt to understand the mechanism of the depolarization process in a fuel cell, an electro-kinetic
Fantastic news! We've Found the answer you've been seeking!
Question:
In an attempt to understand the mechanism of the depolarization process in a fuel cell, an electro-kinetic model for mixed oxygen-methanol current on platinum was developed in the laboratory at FAMU. A very simplified model of the reaction developed suggests a functional relation in an integral form. To find the time required for 50% of the oxygen to be consumed, the time, T(s) is given by
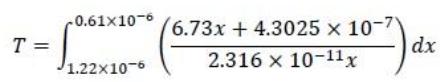
1. Using Trapezoidal rule write a code to find the time required for 50% of the oxygen to be consumed (NB don't use the trapz command).
2. Use the trapz function to solve the above equation and find the time elapsed.
Related Book For 

Management Accounting Information for Decision-Making and Strategy Execution
ISBN: 978-0137024971
6th Edition
Authors: Anthony A. Atkinson, Robert S. Kaplan, Ella Mae Matsumura, S. Mark Young
Posted Date:




