In nitration of acetophenone, an Electrophilic Aromatic Substitution reaction, shown below, m-nitroacetophenone appears to be the...
Fantastic news! We've Found the answer you've been seeking!
Question:
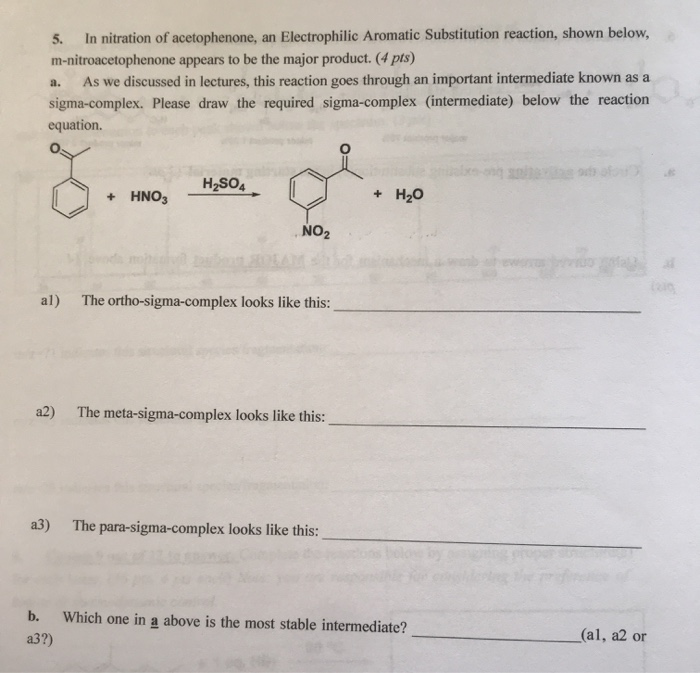
Transcribed Image Text:
In nitration of acetophenone, an Electrophilic Aromatic Substitution reaction, shown below, m-nitroacetophenone appears to be the major product. (4 pts) a. As we discussed in lectures, this reaction goes through an important intermediate known as a sigma-complex. Please draw the required sigma-complex (intermediate) below the reaction equation. 5. + HNO3 H₂SO4 NO₂ al) The ortho-sigma-complex looks like this: a2) The meta-sigma-complex looks like this: a3) The para-sigma-complex looks like this: + H₂O b. Which one in a above is the most stable intermediate? a3?) (al, a2 or (210 In nitration of acetophenone, an Electrophilic Aromatic Substitution reaction, shown below, m-nitroacetophenone appears to be the major product. (4 pts) a. As we discussed in lectures, this reaction goes through an important intermediate known as a sigma-complex. Please draw the required sigma-complex (intermediate) below the reaction equation. 5. + HNO3 H₂SO4 NO₂ al) The ortho-sigma-complex looks like this: a2) The meta-sigma-complex looks like this: a3) The para-sigma-complex looks like this: + H₂O b. Which one in a above is the most stable intermediate? a3?) (al, a2 or (210
Expert Answer:
Answer rating: 100% (QA)
Ans5 a HNO3 HSO4 HNO3 HSO Ortho sigma complex a 10 NO NO... View the full answer

Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
Which compound will undergo an electrophilic aromatic substitution reaction more rapidly, benzene or hexadeuteriobenzene? or D- H.
-
Which of these media executions appears to be the most innovative?
-
Which of these ideas appears to be the most creative use of new technology?
-
Common structural steel can be represented on drawings using a letter, immediately followed by a number, then another number (for example, S14x30). The two numbers for a steel member W16x60 designate...
-
What experiences have you already had in life that would prepare you for leading subordinates through adversity?
-
Following is the adjusted trial balance, with accounts in alphabetical order, for TRN Magazine as at January 31, 2020: Required Prepare the closing entries. Debit Credit $ 21,400 Accounts receivable....
-
The following are six journal entries Keene Engineering, Inc., made during the month of April. Requirement 1. For each transaction shown, determine the accounts affected, the type of account, whether...
-
Tohono Companys 2013 master budget included the following fixed budget report. It is based on an expected production and sales volume of 20,000 units. Required 1. Classify all items listed in the...
-
Here is the region of integration of the integral a. dy dz dx b. dy dx dz c. dx dy dz d. dx dz dy e. dz dx dy 9 81 81-y UT -9x2 0 dz dy dx. Rewrite the integral as an equivalent integral in the...
-
To examine this issue, imagine that banks can charge any borrower 150 basis points above the interest rate at which the bank would break even (in an expected value sense) on that borrower. This is a...
-
Company XYZ is considering an international investment project in a region with a history of political instability. The project is expected to generate annual cash flows of $1,000,000 for the next...
-
On April 1, 20X7, Miller Oil Company purchased a pumping truck. The sole consideration was a \($100\),000 note due in one year. Interest of \($12\),000 was included the face amount of the note. If...
-
Ultrafiltration of a protein solution at constant volume is achieved by the addition of water or buffer to the feed in an operation called diafiltration. Consider a protein diafiltration system where...
-
Indicate whether each of the following statements pertains to microeconomics or macroeconomics: a. The unemployment rate in the United States was 3.7 percent in 2019. b. Workers at the Sleepy Eye...
-
Wild Man Wilson hosts a television show where he gives investment opinions about companies to call-inviewers. Below is the transcript of a portion of one of his shows that focused on employee...
-
Lawrence Bodine is employed by Baylor Health Systems. During the month of June, Lawrence worked 195 hours. 15 of these hours were overtime, and were required to be paid at 150% of the normal hourly...
-
Choose a country china and research if it either collectivist or individualist on each of Hofstede's Cultural Dimensions. Then, complete this for south Korea. compare and contrast Hofstede's Cultural...
-
(a) As Section 17.3 discusses, high-frequency sound waves exhibit less diffraction than low-frequency sound waves do. However, even high-frequency sound waves exhibit much more diffraction under...
-
For each of the following reactions, (1) indicate whether elimination will occur via an E2 or an E1 reaction, and (2) give the major elimination product of each reaction, ignoring stereoisomers: a....
-
The rate of the reaction of 1-bromo-2-butene with ethanol is increased if silver nitrate is added to the reaction mixture. Explain.
-
The 1H NMR spectra of three isomers with molecular formula C7H14O are shown here. Which isomer produces which spectrum? a. b. c. QUESTION CONTINUE TO NEXT PAGE 0 (ppm) frequency 6 (ppm) frequency 8...
-
Expand \((\vec{V} \cdot abla) \vec{V}\) in cylindrical coordinates by direct substitution of the velocity vector to obtain the convective acceleration of a fluid particle. Verify the results given in...
-
Consider a flow field represented by the stream function \(\psi=3 x^{5} y-10 x^{3} y^{3}+3 x y^{5}\). Is this a possible two-dimensional incompressible flow? Is the flow irrotational?
-
Consider the velocity field for flow in a rectangular "corner," \(\vec{V}=A x \hat{i}-A y \hat{i}\), with \(A=0.3 \mathrm{~s}^{-1}\), as in Example 5.8. Evaluate the circulation about the unit square...

Study smarter with the SolutionInn App


