In the following reaction: KMNO4 + NazCz04 + H2SO4 + CO2 + MnSO4 + NazSO4 +...
Fantastic news! We've Found the answer you've been seeking!
Question:
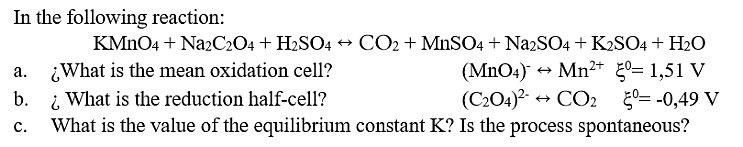
Transcribed Image Text:
In the following reaction: KMNO4 + NazCz04 + H2SO4 + CO2 + MnSO4 + NazSO4 + K2SO4 + H2O ¿What is the mean oxidation cell? i What is the reduction half-cell? What is the value of the equilibrium constant K? Is the process spontaneous? (MnO4) Mn²+ 0= 1,51 V (C204)? + а. b. + CO2 50= -0,49 V с. In the following reaction: KMNO4 + NazCz04 + H2SO4 + CO2 + MnSO4 + NazSO4 + K2SO4 + H2O ¿What is the mean oxidation cell? i What is the reduction half-cell? What is the value of the equilibrium constant K? Is the process spontaneous? (MnO4) Mn²+ 0= 1,51 V (C204)? + а. b. + CO2 50= -0,49 V с.
Expert Answer:
Answer rating: 100% (QA)
clASSMate Solution Date Page The Reaction has two mode ones is oxidation and another reduction both ... View the full answer

Posted Date:
Students also viewed these chemical engineering questions
-
In the following reaction of trichloroacetic acid, HC 2 Cl 3 O 2 , with formate ion, CHO 2 , the formation of trichloroacetate ion, C 2 Cl 3 O 2 , and formic acid, HCHO 2 is favored. Which is the...
-
There is only one value of the equilibrium constant for a particular system at a particular temperature, but there are an infinite number of equilibrium positions. Explain.
-
Which atoms in the following reaction undergo changes in their oxidation state? Which atom is oxidized? Which one is reduced? 2CH3CH2OH + 2Na 2CH3CH2ONa + H2
-
1. Debate whether or not online retailers should be required to collect state sales taxes. Suggest an equitable solution to this issue. 2. Research online tax rules. Look specifically at the 1992...
-
In Exercise 8.31, you found a 95% confidence interval for the mean amount of all venture-capital investments in the fiber optics business sector to be from $5.389 million to $7.274 million. Obtain...
-
Demetria Davis, the bookkeeper for Home Interiors and Designs Company, has just finished posting the closing entries for the year to the ledger. She is concerned about the following balances: Capital...
-
We continue the analysis begun in Exercise 1.7 by examining the relation between y = LIFEEXP and x = FERTILITY, shown in Figure 2.12. Fit a linear regression model of LIFEEXP using the explanatory...
-
Current-carrying capacity of wire, a copper wire of 0.040 in. diameter is insulated uniformly with plastic to an outer diameter of 0.12 in. and is exposed to surroundings at 100F. The heat transfer...
-
a)Name and explain the major Components of a production system E D (6 marks) A 1 B 3 4 4 C Figure 2: Transversing a graph of a travelling salesman b) Use the nearest neighbour heuristic to find the...
-
Situation Russell International, a publicly traded company, reacquired 500,000 shares of its common stock during July 2008 at a cost of $25 per share. The current market price of the stock was $20...
-
How much is the target variable refers to the following data by assuming that fixed costs cannot be reduced? A) $180,000 B) $300,000 C) $220,000 D) $500,000 Target full product cost Actual fixed cost...
-
What are some examples that have caused plaintiff-oriented cases to be looked at negatively?
-
What happens if either party contests the arbitration decision?
-
True Or False The British rule may increase administrative costs of the judicial system.
-
Has the business community benefited from tort reform?
-
What is a fee-shifting rule, and does it curb frivolous lawsuits?
-
John kirk owns a golf course and wants to add computers to the lounge. The cost of computers is 15000, 3 year life, no residual value, expected to generate 12000 annual cash flows, discount rate 8 %....
-
In the figure, two loudspeakers, separated by a distance of d1 = 2.63 m, are in phase. Assume the amplitudes of the sound from the speakers are approximately the same at the position of a listener,...
-
Which set of equipotential surfaces matches this electric field? 111
-
A proton is released from rest at point Q, where the potential is 0 V. Afterward, the proton. A. Remains at rest at Q. B. Moves toward P with a steady speed. C. Moves toward P with an increasing...
-
Rank in order, from largest to smallest, the potential differences V 12 , V 13 , and V 23 between points 1 and 2, points l and 3, and points 2 and 3.

Study smarter with the SolutionInn App

