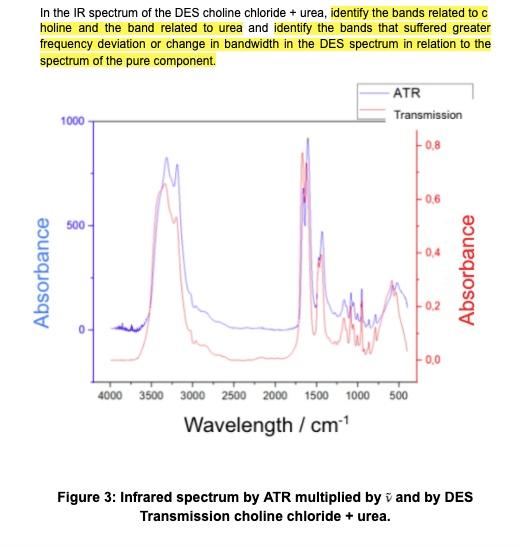
In the IR spectrum of the DES choline chloride + urea, identify the bands related to...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
In the IR spectrum of the DES choline chloride + urea, identify the bands related to c holine and the band related to urea and identify the bands that suffered greater frequency deviation or change in bandwidth in the DES spectrum in relation to the spectrum of the pure component. -ATR Transmission 1000 0,6 500 0,4 0.2 0,0 4000 3500 3000 2500 2000 1500 1000 500 Wavelength / cm1 Figure 3: Infrared spectrum by ATR multiplied by v and by DES Transmission choline chloride + urea. Absorbance Absorbance In the IR spectrum of the DES choline chloride + urea, identify the bands related to c holine and the band related to urea and identify the bands that suffered greater frequency deviation or change in bandwidth in the DES spectrum in relation to the spectrum of the pure component. -ATR Transmission 1000 0,6 500 0,4 0.2 0,0 4000 3500 3000 2500 2000 1500 1000 500 Wavelength / cm1 Figure 3: Infrared spectrum by ATR multiplied by v and by DES Transmission choline chloride + urea. Absorbance Absorbance
Expert Answer:

Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
The IR spectrum of trans-oct-2-enoic acid is shown. Point out the spectral characteristics that allow you to tell that this is a carboxylic acid, and show which features lead you to conclude that the...
-
The IR spectrum of a compound with molecular formula C5H8O was obtained in CCl4 and is shown in Figure 13.42. Identify the compound. Wavelenga qum) 15 16 14 3600 340) 3800 3300 3000 280K 2600 2400...
-
The IR spectrum of propanoic acid (Fig. 2.17) indicates that the absorption for the O-H stretch of the carboxylic acid functional group is due to a hydrogen-bonded form. Draw the structure of two...
-
List 5 Business Uses of Location Analytics? In what way a business can use location data?
-
An appliance manufacturer is considering the purchase of a new machine for stamping out sheet metal parts. If 0 (given) is the true average of the number of good parts stamped out per hour by their...
-
You need to Read the discussion "Lynne Scholz" and give response: No company ever plans on having to deal with a public scandal. Unfortunately for Volkswagen, the actions of a few individuals at a...
-
A hydrogen atom is excited from its ground state to the \(n=\) 4 state. The atom subsequently emits three photons, one of which has a wavelength of \(656 \mathrm{~nm}\). What are the wavelengths of...
-
At December 31, 2010, Percheron Inc. had a deferred tax asset of $30,000. At December 31, 2011, the deferred tax asset is $59,000. The corporations 2011 current tax expense is $61,000. What amount...
-
How unique or innovative packaging can benefit consumers and profit producers. Respond to the following: Identify a company that has used unique and innovative packaging for its products. Describe...
-
For jet aircraft flying out of Houston on a summer afternoon (T = 95 F, 70% relative humidity) and Palm Springs on a winter morning (T = 40 F, 55% relative humidity), determine for both locations: a)...
-
A particle of mass m is moving in a potential of the form, If the particle is at stable equilibrium at a point x,. then x is equal to places). (Upto two decimal
-
The sharing of profits from joint ownership of property is usually enough to create a partnership. (True/False)
-
Employment at will means that employers can fire employees only with good cause. (True/False)
-
What is a share exchange?
-
Cumulative voting allows minority shareholders to obtain representation on the board of directors. (True/False)
-
Midwest Movers, Inc., and Northwest Transport Corporation consolidate. When this combination is completed, a. Midwest and Northwest will cease to exist. b. Midwest will continue as the sole surviving...
-
What is a consequence of Americans' lack of political knowledge? Click or tap a choice to answer the question. It makes citizens more likely to seek political information over the internet. It leads...
-
United Business Forms capital structure is as follows: Debt ............................................ 35% Preferred stock ........................... 15 Common equity .......................... 50...
-
The cyclohexane chair just drawn has the headrest to the left and the footrest to the right. Draw a cyclohexane chair with its axial and equatorial bonds, having the headrest to the right and the...
-
Show how you would accomplish the following conversions in good yields. You may use any necessary reagents. (a) (b) (c) (d) (e) (f) CH, CH,-C-C-CH3 CH3 O CH3 CH Br CH3 CH COOH COOH CH O CH O 0...
-
An important protecting group developed specifically for polyhydroxy compounds like nucleosides is the tetraisopropyldisiloxanyl group, abbreviated TIPDS, that can protect two alcohol groups in a...
-
The state of stress at a point in the \(x y z\) coordinates is Determine the stress matrix relative to the \(x^{\prime} y^{\prime} z^{\prime}\) coordinates, which is obtained by rotating the \(x y...
-
Find the principal stresses and the orientation of the principal axes of stresses for the following cases of plane stress. a. \(\sigma_{x x}=40 \mathrm{MPa}, \sigma_{y y}=0 \mathrm{MPa}, \tau_{x...
-
Determine the principal stresses and their associated directions, when the stress matrix at a point is given by 11 1 [6]=112 MPa. [121]

Study smarter with the SolutionInn App


