In which one of the following species does the transition metal ion have d electron configuration?...
Fantastic news! We've Found the answer you've been seeking!
Question:
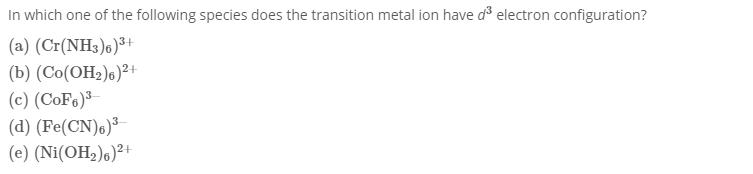
Transcribed Image Text:
In which one of the following species does the transition metal ion have d³ electron configuration? (a) (Cr(NH3)6) ³+ 3+ (b) (Co(OH₂)6)²+ (c) (CoF6)³ (d) (Fe(CN)6)³ (e) (Ni(OH₂)6)²+ In which one of the following species does the transition metal ion have d³ electron configuration? (a) (Cr(NH3)6) ³+ 3+ (b) (Co(OH₂)6)²+ (c) (CoF6)³ (d) (Fe(CN)6)³ (e) (Ni(OH₂)6)²+
Expert Answer:
Answer rating: 100% (QA)
Solution 8 a CrNH3 Cr3 Ar3d D COOH 2 x 60 2 2 Charge in 20 0 ... View the full answer

Related Book For 

Posted Date:
Students also viewed these chemical engineering questions
-
In which one of the following structures does the central atom have a formal charge of +2? a. SF b. SO,- . d. BeCl, e. AICI :: :0: :i: :0 Be :O: :: Select one: a. e b. a . d d. b e. c O O
-
The correct answers 1) A,C 24. Which anion is strongest base? 1) C,H,0- 2) NO, 3) CI 4)CH,CO0- 2) B,D 3) A,D 4) , 25. The strongest base of the following species is 37. Formation of dative bond is...
-
e Let A=d e fand B= 3a 3b 3c Which one of the hi 2a -g 2b-h 2c-i following statements must be true? O A. det B = 6 det A B. det B = -6 det A C. det B = -2 det A O D. det B = 3 det A O E. det B = -3...
-
DrinkOh Limited uses an application service provider to process its payroll. Its employees enter their hours using their smart phones. The payroll clerk collects the smart phone data and transmits it...
-
The number of X-rays taken and X-ray costs over the last nine months in Beverly Hospital are given below: Required: 1. Using the high-low method, estimate the cost formula for X-ray costs. 2. Using...
-
6 Calculate the pressure (in lb/in 2 ) on the hatch of a submarine having an area of 4 ft, if the outside water pressure is 2.5 atmospheres and the pressure inside the cabin is 1 atmosphere. Please...
-
A 10-m-long, 5.042-cm I.D. copper pipe has two fully open gate valves, a swing check valve, and a sudden enlargement to a 9.919-cm I.D. copper pipe. The \(9.919 \mathrm{~cm}\) copper pipe is \(5.0...
-
1. Respond to the following question: Would X be someone whom you would consider as a viable potential candidate for the hospital administrator position, despite his limited technical expertise? 2....
-
The following is a sequence of eight numbers: 01 2 3 4 5 6 7 350 100 250 400 300 50 200 150 Use QuickSort to sort the numbers in the sequence in ascending order. Show the result of each step by...
-
Recall Exercise 5.2.4 where researchers Wilt et al. (New England Journal of Medicine, 2012) investigated whether surgery, compared to just observation, was (more) effective in improving mens survival...
-
A STRIPS traded on April 1, 2023, matures in 18 years on April 1, 2041. Assuming a yield to maturity of 5.8 percent, what is the STRIPS price? Note: Use Excel to answer this question. Enter your...
-
Determine which of the foUowing two companies would be eligible for listing on the NYSE: Firm 1 Firm 2 Earnings before taxes for the last 3 years Value of publicly held stock Number of common shares...
-
Conduct an online estate planning article search on current and future estate tax law changes. Write a brief summary of recommendations from the articles showing how future changes in the tax code...
-
The text of a simple will can be found on the Internet. Locate and print at least two wills. Compare the language and clauses, as described in your text. What are the advantages and disadvantages of...
-
To learn more about "cheap" living, do an Internet search to generate a list of the "top ten tips" for living frugally or saving money. How do the suggested spending and saving practices vary over...
-
Which of these data are quantitative and which are qualitative? a. A countrys unemployment rate. b. A countrys population. c. A countrys gross domestic product (GDP). d. Whether a country belongs to...
-
On January 1, 20X6, KJ Corporation acquired equipment for CU 260,000. The estimated life of the equipment is 5 years. The estimated residual value is CU 20,000. What is the balance in Accumulated...
-
A police officer pulls you over and asks to search your vehicle because he suspects you have illegal drugs inside your car. Since he doesn't have reasonable suspicion to search your car, legally he...
-
The quantity of a radioactive material is often measured by its activity (measured in curies or millicuries) rather than by its mass. In a brain scan procedure, a 70-kg patient is injected with 20.0...
-
The standard enthalpy of atomization of an element is the energy required to convert one mole of an element in its most stable form at 25C to one mole of monatomic gas. Given that the standard...
-
The atomic masses of 63Li and 73Li are 6.0151 amu and 7.0160 amu, respectively. Calculate the natural abundances of these two isotopes. The average atomic mass of Li is 6.941 amu.
-
Amanda Company started business at the beginning of 2022. The company applies the lower-of-cost- or-net-realizable-value rule to its inventory as a whole. Inventory cost and market value as of the...
-
Which one of the following costs is not included in inventory for manufacturing firms? a. Raw materials used in production b. Raw materials unused and stored in the warehouse c. Salary paid to the...
-
Stevenson Company's trial balance as of December 31, 2022 shows \($35,000\) for inventory; but the balance sheet shows \($33,000.\) Assuming no inventory shrinkage, what is the most likely reason for...

Study smarter with the SolutionInn App


