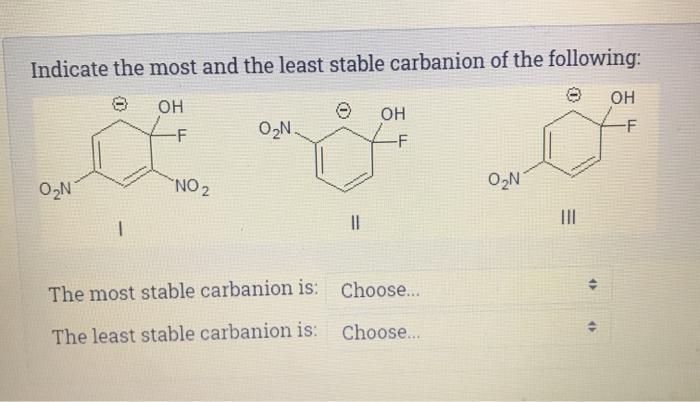
Indicate the most and the least stable carbanion of the following: OH 0N -F NO ...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
Indicate the most and the least stable carbanion of the following: OH 0₂N -F NO ₂ O₂N. || OH -F The most stable carbanion is: Choose... The least stable carbanion is: Choose... O₂N ||| 46 OH -F Indicate the most and the least stable carbanion of the following: OH 0₂N -F NO ₂ O₂N. || OH -F The most stable carbanion is: Choose... The least stable carbanion is: Choose... O₂N ||| 46 OH -F
Expert Answer:
Answer rating: 100% (QA)
compound with more ternary carbon atoms is more sta... View the full answer

Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
The most stable conformation of 5-hydroxy-1,3-dioxane has the OH group in an axial position, rather than an equatorial position. Provide an explanation for this observation.
-
Choose the more stable alkene in each of the following pairs. Explain your reasoning. (a) 1-Methylcyclohexene or 3-methylcyclohexene (b) Isopropenylcyclopentane or allylcyclopentane (c) (d)...
-
The resonance form shown is not the most stable one for the compound indicated. Write the most stable resonance form.
-
Litchfield Industries gathered the following information for the month ended June 31: The static budget volume is 5000 units: Overhead flexible budget: Number of units 8,000 9,000 10,000 Standard...
-
1. Analyze the following transactions, assuming that a business-type activity was involved. 2. Analyze the following transactions, assuming that a general government activity was involved. a....
-
Wilco Corporation has the following account balances at December 31, 2012. Share capitalordinary, $5 par value ....$ 510,000 Treasury shares ..............90,000 Retained earnings...
-
The following scenarios describe situations faced by hypothetical companies that currently have a centralized organization structure. As you review each of the scenarios, provide your opinion as to...
-
Dozier Industries Inc. manufactures only one product. For the year ended December 31, 2014, the contribution margin increased by $38,500 from the planned level of $1,386,000. The president of Dozier...
-
Calculate the following for a 3 ft 4 ft window with the label to the right. The summer design conditions for peak cooling load are 70 and 60% RH inside; 90 and 90% outside; and a peak direct solar...
-
Washington State Fisheries, Incorporated, processes salmon for various distributors. Two departments, processing and packaging, are involved. Data relating to tons of salmon sent to the processing...
-
Which of the following statements is true about Expenses? O They belong on the income statement and show what the company has earned by providing goods or services. O They are earned by the company,...
-
How do leaders navigate the complexities of team dynamics, including conflicts, power struggles, and role ambiguity, while promoting constructive dialogue, conflict resolution, and mutual...
-
Using scholarly resources which do you feel is the most limiting factor in generating innovations: the idea for an innovation, or the organizational processes to support innovation? Why? Do you think...
-
Briefly describe and analyze the relationship between emotional intelligence and conflict resolution strategies within high-stress work settings?
-
What are the implications of cross-cultural communication barriers for multinational corporations in terms of team collaboration and conflict resolution?
-
Create and evaluate a scenario where, according to the Maryland Attorneys Rules of Professional Conduct, a conflict of interest might exist. The scenario must be detailed with names, actions, and a...
-
The spread of the bank is 1.00%, the cost rate on funds is 6.00%. If the banks' earning assets are $1,500,000 and Interest bearing Liabilities are $1,200,000, then the net interest income of the bank...
-
The bookkeeper for Riley, Inc., made the following errors: a. A cash purchase of supplies of $357 was recorded as a debit to Supplies for $375 and a credit to Cash of $375. b. A cash sale of $3,154...
-
Draw the resonance contributors for the carbocation intermediates obtained from electrophilic aromatic substitution at the 1-position and the 2-position of naphthalene. Use the resonance contributors...
-
Give two sets of alkyl bromide and alkene that could be used in a Heck reaction to prepare the following compound: CH3C CH= CH OCH3
-
Determine the structure of each of the following compounds based on its mass, IR, and 1H NMR spectra. a. b. 100 u 50 27 114 20 40 60 80 100 120 m/z 3 2 2 28 2.9 10 13 14 15 6 6 5 2 (ppm) frequency...
-
Gross profit is (A) Excess of sales over cost of goods sold (B) Sales less purchases (C) Cost of goods sold + opening inventory (D) Net profit less expenses of the period
-
To find the value of closing inventory at the end of a period we (A) Do this by physically counting the inventory (i.e. stocktaking) (B) Look in the inventory account (C) Deduct opening inventory...
-
Net profit is calculated in the (A) Trading account (B) Profit and loss account (C) Trial balance (D) Statement of financial position

Study smarter with the SolutionInn App


