INFO:: Used 3 ml styreNE 0.40 ml initiator tert butyl obtained 0.8660 g polystyrene polymerized 1.5512 g
Fantastic news! We've Found the answer you've been seeking!
Question:



INFO:: Used 3 ml styreNE
0.40 ml initiator tert butyl
obtained 0.8660 g polystyrene
polymerized 1.5512 g styrene
QUESTIONS:
1. A feature of polymers compared to “normal” molecules, which have fixed molecular weights, is that the same polymer can have a range of molecular weights depending on the conditions under which it was made. For the reaction we did, assume each initiator molecule starts one polystyrene chain and each chain is the same length, calculate the number of styrene molecules per chain and thus the weight of a chain of YOUR polymer.
2. Suggest a reason why methanol (the solvent used in this lab) is a better solvent for isolating polystyrene as a powder than acetone.
Transcribed Image Text:
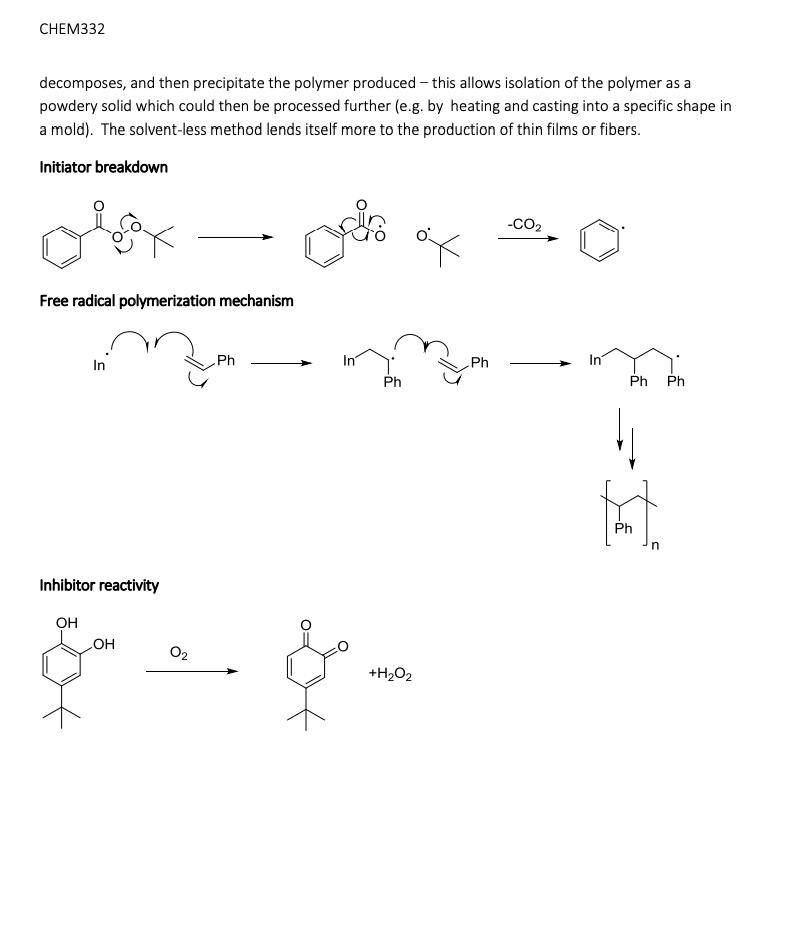
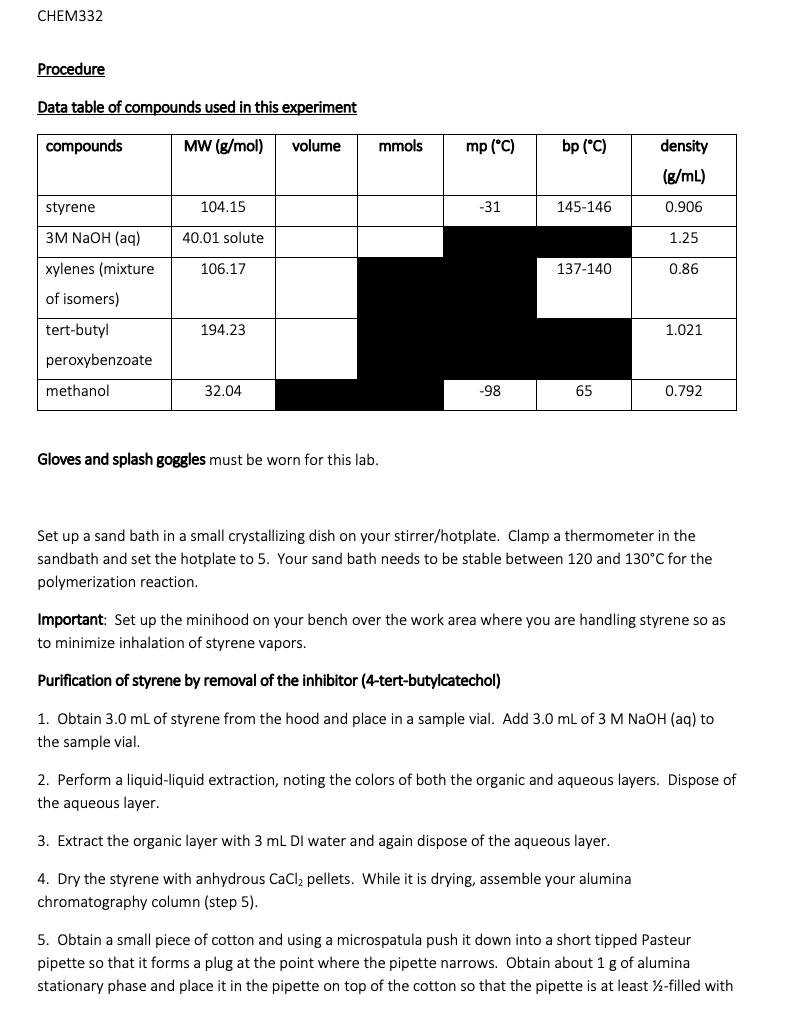
Simple alkenes are one of the most common types of monomer for polymerization reactions. Although they can be polymerized by ionic mechanisms, free radical polymerization is the most common method and you may have discussed the mechanism in CHEM331. In this lab we will polymerize styrene to form polystyrene, which is made on a huge scale industrially every year (millions of tonnes): Its primary uses are in packaging materials and in containers. One major issue associated with polystyrene is environmental - the material is very stable due to the strong, non-polar covalent bonds that comprise the alkane polymer chain and so is very slow to break down in the environment (biodegrade). This leads to build up of plastics in the ocean and other areas if it is not disposed of properly. Free radical polymerization requires an initiator - a compound that breaks apart easily to form free radicals that then begin the chain growth process. Initiators are usually compounds that contain a weak covalent bond that can be readily homolytically cleaved with either heat or light to form radicals: We will be using tert-butyl peroxybenzoate which contains a weak peroxy, or -0-0-, bond. OH Ph or tert-butyl peroxybenzoate OH 4-tert-butyl catechol styrene Many alkenes are so reactive towards this type of polymerization that the reaction may be initiated just by the presence of O₂ (itself a radical). To prevent this, the monomer must be stored long term at low temperatures and in the presence of an inhibitor, a compound which preferentially reacts with O₂ thus protecting the monomer. The amount of inhibitor is quite small, in the range 10-15 ppm. In this experiment, we will first remove the inhibitor from the styrene monomer before attempting the polymerization reaction. This is easily done as the inhibitor is 4-tert-butyl catechol, which contains weakly acidic phenol groups, and so reaction with a strong base and liquid-liquid extraction allows removal of the deprotonated form in the aqueous layer (see the acid-neutral separation lab from CHEM331). Polymerization of alkenes can be done in the absence or presence of a solvent. In this lab we will do the polymerization of styrene in the solvent xylenes (dimethylbenzene) at reflux so that the initiator thermally Simple alkenes are one of the most common types of monomer for polymerization reactions. Although they can be polymerized by ionic mechanisms, free radical polymerization is the most common method and you may have discussed the mechanism in CHEM331. In this lab we will polymerize styrene to form polystyrene, which is made on a huge scale industrially every year (millions of tonnes): Its primary uses are in packaging materials and in containers. One major issue associated with polystyrene is environmental - the material is very stable due to the strong, non-polar covalent bonds that comprise the alkane polymer chain and so is very slow to break down in the environment (biodegrade). This leads to build up of plastics in the ocean and other areas if it is not disposed of properly. Free radical polymerization requires an initiator - a compound that breaks apart easily to form free radicals that then begin the chain growth process. Initiators are usually compounds that contain a weak covalent bond that can be readily homolytically cleaved with either heat or light to form radicals: We will be using tert-butyl peroxybenzoate which contains a weak peroxy, or -0-0-, bond. OH Ph or tert-butyl peroxybenzoate OH 4-tert-butyl catechol styrene Many alkenes are so reactive towards this type of polymerization that the reaction may be initiated just by the presence of O₂ (itself a radical). To prevent this, the monomer must be stored long term at low temperatures and in the presence of an inhibitor, a compound which preferentially reacts with O₂ thus protecting the monomer. The amount of inhibitor is quite small, in the range 10-15 ppm. In this experiment, we will first remove the inhibitor from the styrene monomer before attempting the polymerization reaction. This is easily done as the inhibitor is 4-tert-butyl catechol, which contains weakly acidic phenol groups, and so reaction with a strong base and liquid-liquid extraction allows removal of the deprotonated form in the aqueous layer (see the acid-neutral separation lab from CHEM331). Polymerization of alkenes can be done in the absence or presence of a solvent. In this lab we will do the polymerization of styrene in the solvent xylenes (dimethylbenzene) at reflux so that the initiator thermally
Expert Answer:
Answer rating: 100% (QA)
Detailed Explanation The experiment involves the use of tertbutyl peroxybenzoate as an initiator for free radical polymerization The process also includes the removal of the inhibitor 4tertbutyl catec... View the full answer

Related Book For 

Posted Date:
Students also viewed these chemical engineering questions
-
A reality of communication is that the same sign often means different things to different people. The red ribbon, for example, means different things to different groups. Provide an example from...
-
Polystyrene can be made more rigid by copolymerizing styrene with divinylbenzene, What purpose does the divinylbenzene serve? Why is the copolymer more rigid? CH=CH2 CH=CH,
-
Polymer Molecular Weight Distribution the following data were obtained for polyethylene. Determine the average molecular weight and degree of polymerization.
-
Two pulses A and B are moving in opposite directions along a taut string with a speed of 2.00 cm/s. The amplitude of A is twice the amplitude of B. The pulses are shown in Figure P18.2 at t = 0....
-
The high-growth, market-share policy should not bother any franchisee. It simply creates opportunities to invest in more restaurants. Evaluate this statement. In 1980 Wayne Kilburn and his wife, Mary...
-
Write to Oriental Bank of Commerce, Civil Lines, Bhopal, to make enquiries about the credit standing of Messrs. Rajpal Brothers, Raja Bazar, Bhopal, for a sum of Rs.5,00,000. The party's bankers are...
-
Discuss under what circumstances parental consent for a minor might not be necessary.
-
A pistoncylinder device contains 0.85 kg of refrigerant- 134a at 10°C. The piston that is free to move has a mass of 12 kg and a diameter of 25 cm. The local atmospheric pressure is 88 kPa. Now,...
-
25 1 QS 2-7 (Algo) Computing 1-account balance LO CZ Determine the ending balance of each of the following T-accounts. Cash Accounts Payable oints 300 250 4,000 14,000 500 260 6,500 eBook 220 Hint...
-
Using lab 7 with a three-day lead time (all original values), we will analyze six different scenarios to determine which would be the best scenario based on the total cost. Scenario A - Min is 3,500...
-
Explain the specific strategies businesses use to mine new customers and gain competitive advantage through social media. What privacy issues emerge when you review these techniques? As an IT...
-
GIVE COMPREHENSIVE EXAMPLES OF HOW INACCURATE,INCOMPLETE,OR OUTDATED DATA LEAD TO MEDICAL ERRORS,DELAYS IN TREATMENT OR OTHER PROBLEMS
-
What industry standards were used for the basis of the audit? explain all industry standards in detail with examples.
-
If the percentage change in net income changes faster than the percentage change in revenues, what is happening in the business?
-
What role do historical and comparative analyses play in uncovering the underlying mechanisms and contingencies that have shaped the evolution of social structures across different societies and...
-
To what extent do Marxist analyses of social structure emphasize the role of economic relations and class dynamics in shaping social hierarchies, power dynamics, and patterns of exploitation within...
-
1) What is the average return for Home Depot, Inc. (HD)? 2) What is the standard deviation for Costo Wholesale Corporation (COST)? 3)What is the correlation between BBY and MSFT? 4) What is the beta...
-
Find the image of x = k = const under w = 1/z. Use formulas similar to those in Example 1. y| y = 0 -21 -2 -1 -1, /1 12 T -1 -1 y= -2 x =0
-
The hydrolysis of the ester shown here is catalyzed by morpholine, a secondary amine. Propose a mechanism for this reaction. (The pKa of the conjugate acid of morpholine is 9.3, so morpholine is too...
-
The most stable form of glucose (blood sugar) is a six-membered ring in a chair conformation with its five substituents all in equatorial positions. Draw the most stable form of glucose by putting...
-
An aldol addition can be catalyzed by acids as well as by bases. Propose a mechanism for the acid-catalyzed aldol addition of propanal.
-
If you look at the light spectrum reflected from the surface of a DVD compared to the spectrum reflected from the surface of a CD, youll see that the rainbows from the DVD are more spread out, with...
-
Increasing the density of a material tends to increase the index of refraction. Does light travel faster in seawater or in fresh water?
-
Antireflection coatings for glass usually have an index of refraction that is less than that of glass. Explain how this permits a thinner coating.

Study smarter with the SolutionInn App


