Normal rain has a pH of 5.6 due to dissolved atmospheric carbon dioxide at a current...
Fantastic news! We've Found the answer you've been seeking!
Question:

Transcribed Image Text:
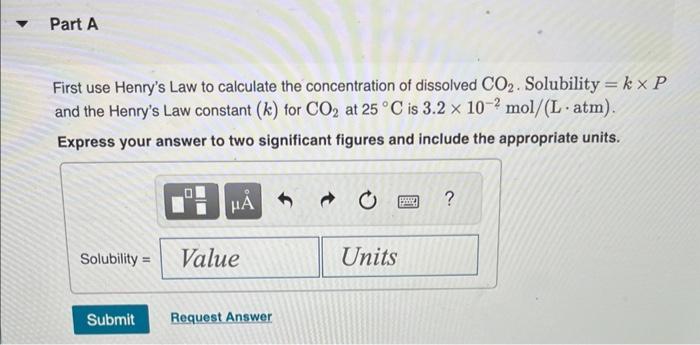
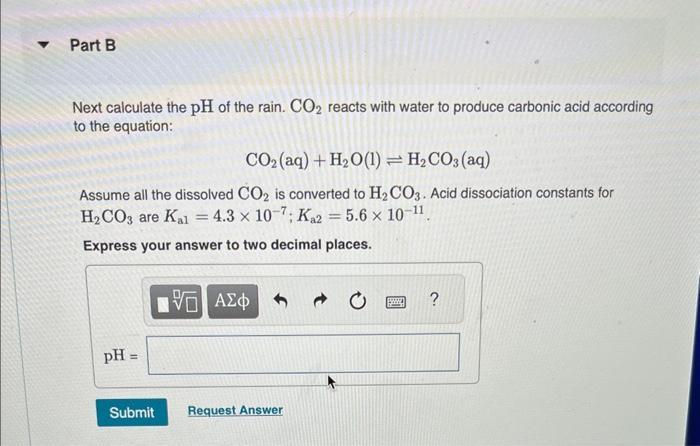
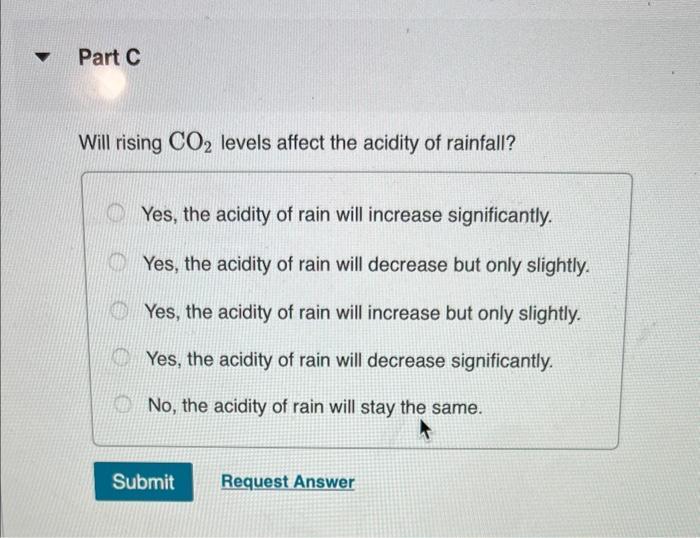
Normal rain has a pH of 5.6 due to dissolved atmospheric carbon dioxide at a current level of 400 ppm. Various models predict that burning fossil fuels will increase the atmospheric CO₂ concentration to between 500 and 1000 ppm (by volume) by the year 2100. Calculate the pH of rain in a scenario where the CO₂ concentration is 550 ppm. Part A First use Henry's Law to calculate the concentration of dissolved CO₂. Solubility = kx P and the Henry's Law constant (k) for CO2 at 25 °C is 3.2 x 10-2 mol/(L atm). Express your answer to two significant figures and include the appropriate units. I Submit HA Solubility= Value Request Answer C Units P ? Part B Next calculate the pH of the rain. CO2 reacts with water to produce carbonic acid according to the equation: CO₂ (aq) + H₂O(1) = H₂CO3(aq) Assume all the dissolved CO₂ is converted to H₂CO3. Acid dissociation constants for H₂CO3 are Kal 4.3 x 10-7; K₁2 = 5.6 x 10-11. Express your answer to two decimal places. pH = Submit 15. ΑΣΦΑ Request Answer → www Part C Will rising CO₂ levels affect the acidity of rainfall? Yes, the acidity of rain will increase significantly. Yes, the acidity of rain will decrease but only slightly. Yes, the acidity of rain will increase but only slightly. Yes, the acidity of rain will decrease significantly. No, the acidity of rain will stay the same. Submit Request Answer Normal rain has a pH of 5.6 due to dissolved atmospheric carbon dioxide at a current level of 400 ppm. Various models predict that burning fossil fuels will increase the atmospheric CO₂ concentration to between 500 and 1000 ppm (by volume) by the year 2100. Calculate the pH of rain in a scenario where the CO₂ concentration is 550 ppm. Part A First use Henry's Law to calculate the concentration of dissolved CO₂. Solubility = kx P and the Henry's Law constant (k) for CO2 at 25 °C is 3.2 x 10-2 mol/(L atm). Express your answer to two significant figures and include the appropriate units. I Submit HA Solubility= Value Request Answer C Units P ? Part B Next calculate the pH of the rain. CO2 reacts with water to produce carbonic acid according to the equation: CO₂ (aq) + H₂O(1) = H₂CO3(aq) Assume all the dissolved CO₂ is converted to H₂CO3. Acid dissociation constants for H₂CO3 are Kal 4.3 x 10-7; K₁2 = 5.6 x 10-11. Express your answer to two decimal places. pH = Submit 15. ΑΣΦΑ Request Answer → www Part C Will rising CO₂ levels affect the acidity of rainfall? Yes, the acidity of rain will increase significantly. Yes, the acidity of rain will decrease but only slightly. Yes, the acidity of rain will increase but only slightly. Yes, the acidity of rain will decrease significantly. No, the acidity of rain will stay the same. Submit Request Answer
Expert Answer:
Answer rating: 100% (QA)
Part A Henrys Law allows us to figure out how much dissolved CO2 is in the rainfall Per Henrys Law solubility is equal to k P Where The concentration ... View the full answer

Related Book For 

Fundamentals of Corporate Finance
ISBN: 978-0077861629
8th edition
Authors: Richard Brealey, Stewart Myers, Alan Marcus
Posted Date:
Students also viewed these chemical engineering questions
-
Comprehensive Problem 1 - Part 1: Taxpayer information, Form 1040, Schedules 1, 2, 3 and 4, Schedule A, and Schedule B. Noah and Joan Arc's Tax Return Note: This problem is divided into three parts....
-
To live comfortably in retirement, you decide you will need to save $2 million by the time you are 65 (you are 30 years old today). You will start a new retirement savings account today and...
-
You believe you will need to have saved $500,000 by the time you retire in 40 years in order to live comfortably. You also believe that you will inherit $100,000 in 10 years. If the interest rate is...
-
The 10-year Coupon Bond has a face value of $1,000, the annual coupon rate is 5 percent (out of its face value), the yield to maturity is 10 percent. (2.a) show me the cash flows of this coupon bond,...
-
First Company sold the following assets during the year. Indicate whether First Company can use the installment method to report each transaction. If not, how is the transaction reported? Assume...
-
(a) Show that a typical drift velocity is of the order of 10 5 cm/s for electrons in a solid subject to an electric field of 5 V over a distance of 1 cm, for around 10 11 s as calculated. (b) Plot...
-
Bonsai Boards income statement data for the year ended December 31,2010, follow. Assume that the ending inventory was accidentally overstated by $3,300. What are the correct amounts for cost of goods...
-
Jones, having filed locally an affidavit required under the assumed name statute, has been operating and advertising his exclusive toy store for twenty years in Centerville, Illinois. His advertising...
-
The input circuit consists of sensors and switches. These signal data to the PCM. Sensors and switches can be divided into "Command" and "Monitoring" devices. Explain what "Command" and "Monitoring"...
-
Imagine that you are Magna's new corporate controller and answer the following: 1. Describe Magna's strategy in terms of how it competes for customers. 2. Based on Magna's strategy and the data...
-
Figure 1 shows a system with a cart with a mass on it. Do the following: a) Find the transfer function . Do not plug in any numerical values yet. X2(5) F(S) b) For the remainder of the question, use...
-
Urn I contains seven black and three white balls; urn II contains twelve black, five white, and six red balls; and urn III contains one black, eight white, and four red balls. Roll a fair die. If the...
-
Write down a MatLab command that determines the FIR lattice coefficients from the FIR direct-form coefficients.
-
Suppose there is a structure that is free from zero-input limit cycles, when employing magnitude truncation, and that it is also forced-input stable when using saturation arithmetic. Discuss whether...
-
By replacing \(z\) with \(-z\) in an RRS filter, where will its pole and zeros be located? What type of magnitude response will result?
-
Use the information given in Problem 24 to find: (a) P(A 1 |E) (b) P(A 2 |E) (c) P(A 3 |E) Data from Problem 24 Suppose that events A 1 , A 2 , and A 3 form a partition of the sample space S with P(A...
-
Outline any five differences between the following approaches to testing in education: I. Norm referenced vs Criterion referenced assessments II. Formative vs summative evaluations
-
I frequently use NY Times and CNN and am aware of Fox News but I never use it. I visit these sites, NY Times and CNN, a few times a week whenever I have to research something or see something on...
-
Percentage of sales models usually assume that costs, fixed assets, and working capital all increase at the same rate as sales. When do you think that these assumptions do not make sense? Would you...
-
Common Products has just made its first issue of stock. It raised $2 million by selling 200,000 shares of stock to the public. These are the only shares outstanding. The par value of each share was...
-
Modern Artifacts can produce keepsakes that will be sold for $80 each. Nondepreciation fixed costs are $1,000 per year, and variable costs are $60 per unit. The initial investment of $3,000 will be...
-
Fill in the Blank. In the finite element method, \(a(n)\) ___________ solution is assumed within each element.
-
The stiffness matrix of a bar element is given by a. \(\frac{E A}{l}\left[\begin{array}{ll}1 & 1 \\ 1 & 1\end{array} ight]\) b. \(\frac{E A}{l}\left[\begin{array}{rr}1 & -1 \\ -1 & 1\end{array}...
-
What is the basis for the derivation of transformation matrices?

Study smarter with the SolutionInn App


