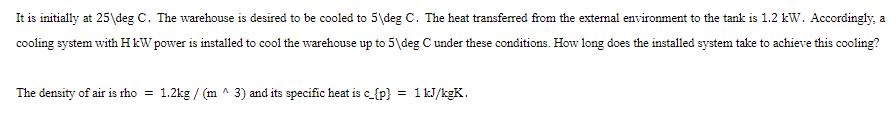It is initially at 25deg C. The warehouse is desired to be cooled to 5deg C....
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
It is initially at 25\deg C. The warehouse is desired to be cooled to 5\deg C. The heat transferred from the external environment to the tank is 1.2 kW. Accordingly, a cooling system with H kW power is installed to cool the warehouse up to 5\deg C under these conditions. How long does the installed system take to achieve this cooling? The density of air is rho = 1.2kg / (m^ 3) and its specific heat is c_{p} = 1 kJ/kgK. It is initially at 25\deg C. The warehouse is desired to be cooled to 5\deg C. The heat transferred from the external environment to the tank is 1.2 kW. Accordingly, a cooling system with H kW power is installed to cool the warehouse up to 5\deg C under these conditions. How long does the installed system take to achieve this cooling? The density of air is rho = 1.2kg / (m^ 3) and its specific heat is c_{p} = 1 kJ/kgK.
Expert Answer:
Answer rating: 100% (QA)
SOLUTION To calculate the time it takes for the cooling system to achieve the desired temperature of ... View the full answer

Related Book For 

Posted Date:
Students also viewed these economics questions
-
a. Example 13-1: Batch Reactor with an Exothermic Reaction Wolfram 1. Adiabatic Case: Use Wolfram to see whether you can find a trajectory that is ready to ignite and whose trajectory looks like a...
-
Managing Scope Changes Case Study Scope changes on a project can occur regardless of how well the project is planned or executed. Scope changes can be the result of something that was omitted during...
-
(1.71, 2.05) Use the confidence interval to find the margin of error and the sample mean.
-
Portions of the financial statements for Hawkeye Company are provided below. Required: 1. Prepare the cash flows from operating activities section of the statement of cash flows for Hawkeye Company...
-
To determine the equilibrium distribution \(f(\sigma)\), we minimize the free en\(\operatorname{ergy}(E-T S)\) of the system under the obvious constraint \(\sum_{\sigma} f(\sigma)=1\). For this, we...
-
State the advantages of high-pressure boilers. Explain the construction and working of Babcock and Wilcox boiler with a neat sketch.
-
A population proportion is .58. Suppose a random sample of 660 items is sampled randomly from this population. a. What is the probability that the sample proportion is greater than .60? b. What is...
-
Fortune Software Corp. has assembled the following data for the years ending December 31, 2021 and 2020. (Click the icon to view the current accounts.) Requirement (Click the icon to view the...
-
Orion Controls is a leading manufacturer of industrial valve systems, and Nathan Armstrong, head of Marketing, had been contacted by Andre Gide, EVP of Avion Chemical to place an order for 50 of...
-
Select the true statement from the following: A. There are only two accounting standards in the world: US GAAP and IFRS B. Many countries have their own accounting standards, but US GAAP is the only...
-
Compare Alphabet Inc. and Amazon in Revenue Growth from 2018 to 2021? explain with references.
-
Evaluate (8) (9) 5x+6ydydx =
-
23. Given the following HTML mark-up and CSS rules, what is the text colour of the word Rainy? HTML mark-up: Today is a Rainy Day CSS rules: body {color: black; } section, p { color: green; } p...
-
4 (a): An investment plan in perpetual bond is offering an expected return of Rs. 7500 at the end of every 3 months period. If Sheraz requires a nominal rate of return of 12% per year compounded...
-
Lions Furniture received an invoice in the amount of $6,951 dated September 16, 2021 with terms 5/10, 2/20, n/60. If the invoice is to be paid in full on September 27, how much will the payment be?
-
Background: Businesses take inventory of items for sale for several reasons. For income tax reporting. Inventory is needed to calculate cost of goods sold on a business tax form. Inventory costs...
-
Write a function that reads a Float24_t value: Float24_t float24_read(void) A legitimate float24 value string is of the form: "mantissabexponent" where the mantissa (m) and the exponent (e) may have...
-
Example 11-3: Adiabatic Liquid-Phase Isomerization of Normal Butane. Can you explain why the CSTR volume is smaller than the PFR volume? Example 11-3 Normal butane, C 4 H 10 , is to be isomerized to...
-
The following E(t) curve was obtained from a tracer test on a reactor. E(t)=0.25t04 t in minutes, and E(t) in min 1 . The conversion predicted by the tanks-in-series model for the isothermal...
-
1. What is the NFPA diamond and why is it important? 2. Go to Wikipedia and search ethylene oxide to find the NFPA diamond numbers. 3. Draw the NFPA diamond for the chemicals propylene, isohexane,...
-
Find the two eigenvalues of an operator associated with a \(2 \times 2\) matrix with arbitrary elements.
-
Discretize the product of two functions, as compared to discretizing each function independently, and describe what that means in the language of kets.
-
Consider the unitary operator \(e^{i \hat{A}}\), with \(\hat{A}\) Hermitian and acting on function space. Can it be diagonalized? If so, write an expression for its diagonal elements.

Study smarter with the SolutionInn App