Question: Steam systems are used to generate power, and provide heat energy. They are found in power plants, chemical plants, refineries, and here at the
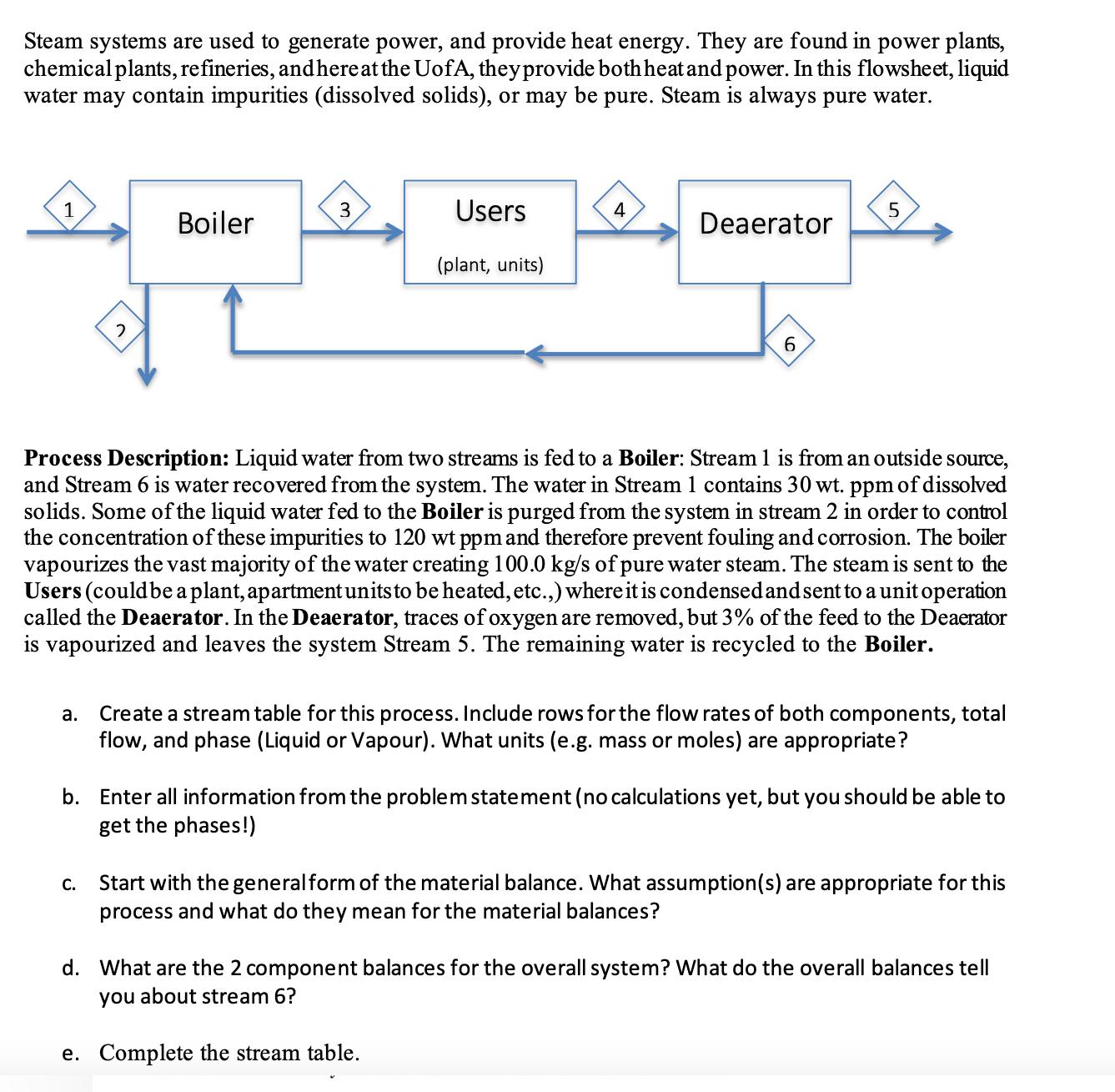
Steam systems are used to generate power, and provide heat energy. They are found in power plants, chemical plants, refineries, and here at the UofA, they provide both heat and power. In this flowsheet, liquid water may contain impurities (dissolved solids), or may be pure. Steam is always pure water. 1 3 Boiler 2 Users 4 5 Deaerator (plant, units) 6 Process Description: Liquid water from two streams is fed to a Boiler: Stream 1 is from an outside source, and Stream 6 is water recovered from the system. The water in Stream 1 contains 30 wt. ppm of dissolved solids. Some of the liquid water fed to the Boiler is purged from the system in stream 2 in order to control the concentration of these impurities to 120 wt ppm and therefore prevent fouling and corrosion. The boiler vapourizes the vast majority of the water creating 100.0 kg/s of pure water steam. The steam is sent to the Users (could be a plant, apartment units to be heated, etc.,) where it is condensed and sent to a unit operation called the Deaerator. In the Deaerator, traces of oxygen are removed, but 3% of the feed to the Deaerator is vapourized and leaves the system Stream 5. The remaining water is recycled to the Boiler. a. Create a stream table for this process. Include rows for the flow rates of both components, total flow, and phase (Liquid or Vapour). What units (e.g. mass or moles) are appropriate? b. Enter all information from the problem statement (no calculations yet, but you should be able to get the phases!) C. Start with the general form of the material balance. What assumption(s) are appropriate for this process and what do they mean for the material balances? d. What are the 2 component balances for the overall system? What do the overall balances tell you about stream 6? e. Complete the stream table.
Step by Step Solution
3.37 Rating (153 Votes )
There are 3 Steps involved in it

Get step-by-step solutions from verified subject matter experts


