The entropy H of a 4-state system is given by: H(p1.P2, P3. Pa) = -EP Inp...
Fantastic news! We've Found the answer you've been seeking!
Question:
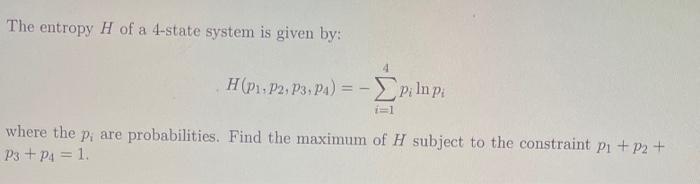
Transcribed Image Text:
The entropy H of a 4-state system is given by: H(p1.P2, P3. Pa) = -EP Inp i i=1 where the p, are probabilities. Find the maximum of H subject to the constraint pi + p2 + P3 + P4 = 1. The entropy H of a 4-state system is given by: H(p1.P2, P3. Pa) = -EP Inp i i=1 where the p, are probabilities. Find the maximum of H subject to the constraint pi + p2 + P3 + P4 = 1.
Expert Answer:
Answer rating: 100% (QA)
Guven thal H and Pit P Ps PA 1 A 3D We use the method of L... View the full answer

Posted Date:
Students also viewed these mathematics questions
-
A first-order system is given by with the initial condition x(0) = .v0. We want to design a feedback controller u = -kx such that the performance index is minimized. (a) Let A = 1. Develop a formula...
-
The impulse response of an LTI system is given by h(n) = (n) (n k0). To determine the impulse response g(n) of the inverse system, an engineer computes the determine N-point DFT H (k), N = 4k0, of...
-
The Hamiltonian operator for a 2-state system is given by H = a1|1){1| + a2|2) (2| + b (|1) (2| + |2) (1|) , where a1, az and b are real numbers, and the kets |1) and |2) form an orthonormal basis....
-
CMOS Chips is hedging a 20-year, $21 million, 8% bond payable with a 20-year interest rate swap and has designated the swap as a fair value hedge. The agreement called for CMOS to receive payment...
-
Identify two competing organizations (for example, AT&T and Verizon, Taylor Made and Callaway golf club manufacturers, or Starbucks and McDonalds). Explain the differences in their missions,...
-
A borrower has two alternatives for a loan: (1) Issue a $75,000, 90-day, 7% note or (2) Issue a $75,000, 90-day note that the creditor discounts at 7%. a. Calculate the amount of the interest expense...
-
The Golden Pairs Company operates a pension scheme that offers defined benefit pensions for the benefit of the companys employees and their spouses. At the end of the reporting period, the present...
-
United Rental Company reported the following information related to its pension plan for the years 20112014. The fund is administered by a separate outside trustee. Instructions: 1. Prepare the...
-
(d) For the following Boolean expression, give: i. The truth table F(a, b, c, d) = ( + b.d).(c.b.a. + .d) ii. The Karnaugh map iii. The minimal sum of products expression
-
Find and analyze at least three sets of proposed principles for the governance of AI. What do the sets of principles have in common? How do they differ? How implementable are these principles?
-
Which of the following is a difference between the U.S. GAAP and IFRS requirements related to inventory? (Select one or more) a. A variety of inventory costing methodologies in permitted in US GAAP....
-
1. Identify an entrepreneur in your area you would like to interview. 2. Contact the person you have selected and make an appointment. Be sure to explain why you want the appointment and to give a...
-
If you had been treated with respect, this would never have happened, would it?
-
Your instructor will divide your class randomly into groups of four to six people. Acting as a team, with everyone offering ideas and one person serving as official recorder, each group will be...
-
Make decisions in the situations described in the Ethical Behavior Worksheet. You will not have all the background information on each situation; instead, you should make whatever assumptions you...
-
1. Identify 3 students to play the roles of the employees. Ask these 3 individuals to read their roles below. 2. Identify 1 student to play the role of the president of the social enterprise (Taylor...
-
Define/describe the following organizations and summarize how they differ from a social enterprise: i) Non-Profit organization iii) Social Venture iii) Charity organization iv) Benefit Company v)...
-
Write each fraction as a percent. 7 50
-
A blender does 5000 J of work on the food in its bowl. During the time the blender runs, 2000 J of heat is transferred from the warm food to the cooler environment. What is the change in the thermal...
-
Which system contains more atoms: 5 mol of helium (A = 4) or 1 mol of neon (A = 20)? A. Helium B. Neon C. They have the same number of atoms.
-
What is the ratio T f /T i for this process? A. 1/4 B. 1/2 C. 1 (no change) D. 2 E. 4 F. There is not enough information to decide. p (atm) 4 3- 2 1 0- 0 2 -V (m)

Study smarter with the SolutionInn App

