Question: a) Calculate the Gibbs free energy (AG) of above reaction (last two digit of your student number 10) C? b) Calculate the equilibrium constant
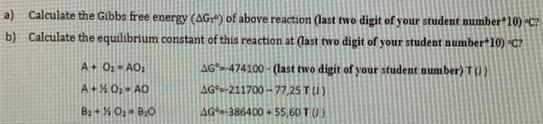
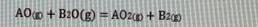
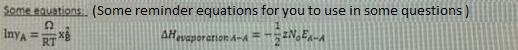
a) Calculate the Gibbs free energy (AG) of above reaction (last two digit of your student number 10) C? b) Calculate the equilibrium constant of this reaction at (last two digit of your student number 10) C? A+ O AO AG-474100 - (last two digit of your student nunmber) T(1) A+X 0: AO AG-211700 -77,25 TU) Ba + % O: BO AG-386400 + 55,60 T () AOo + B20(g) = AOzp + B20 Some equations: (Some reminder equations for you to use in some questions) InyA = RT AHvaporarion A-A
Step by Step Solution
3.50 Rating (153 Votes )
There are 3 Steps involved in it
a 10... View full answer

Get step-by-step solutions from verified subject matter experts
Document Format (2 attachments)
635f2ca77764a_231268.pdf
180 KBs PDF File
635f2ca77764a_231268.docx
120 KBs Word File


