Malonyl-CoA is an intermediate in fatty acid synthesis. It also regulates fatty acid metabolism. Which of...
Fantastic news! We've Found the answer you've been seeking!
Question:
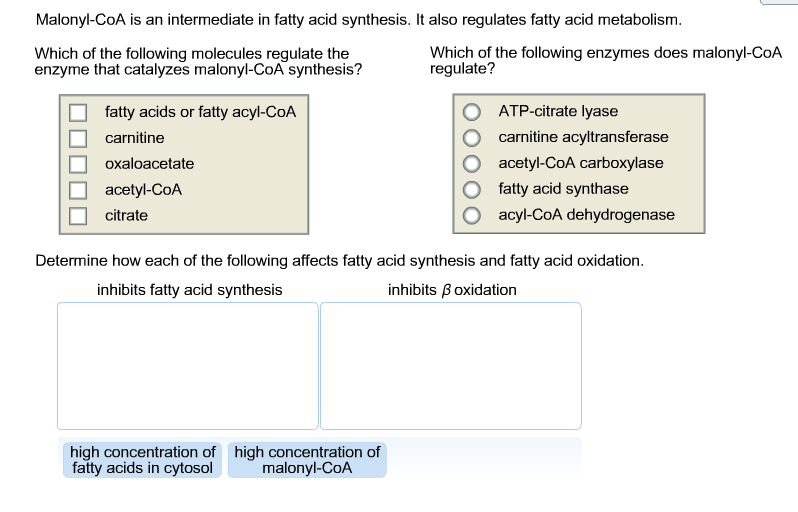
Transcribed Image Text:
Malonyl-CoA is an intermediate in fatty acid synthesis. It also regulates fatty acid metabolism. Which of the following molecules regulate the enzyme that catalyzes malonyl-CoA synthesis? fatty acids or fatty acyl-CoA carnitine oxaloacetate acetyl-CoA citrate Which of the following enzymes does malonyl-CoA regulate? high concentration of high concentration of fatty acids in cytosol malonyl-CoA ATP-citrate lyase carnitine acyltransferase acetyl-CoA carboxylase fatty acid synthase acyl-CoA dehydrogenase Determine how each of the following affects fatty acid synthesis and fatty acid oxidation. inhibits fatty acid synthesis inhibits oxidation Malonyl-CoA is an intermediate in fatty acid synthesis. It also regulates fatty acid metabolism. Which of the following molecules regulate the enzyme that catalyzes malonyl-CoA synthesis? fatty acids or fatty acyl-CoA carnitine oxaloacetate acetyl-CoA citrate Which of the following enzymes does malonyl-CoA regulate? high concentration of high concentration of fatty acids in cytosol malonyl-CoA ATP-citrate lyase carnitine acyltransferase acetyl-CoA carboxylase fatty acid synthase acyl-CoA dehydrogenase Determine how each of the following affects fatty acid synthesis and fatty acid oxidation. inhibits fatty acid synthesis inhibits oxidation
Expert Answer:
Answer rating: 100% (QA)
Which of the following molecules regulate the enzyme that catalyzes malonylCoA synthesis acetylC... View the full answer

Related Book For 

Organic Chemistry
ISBN: 978-1118875766
12th Edition
Authors: T. W. Graham Solomons, Craig B. Fryhle, Scott A. Snyder
Posted Date:
Students also viewed these chemistry questions
-
Which of the following molecules would you expect to be aromatic? (a) (b) (c) (d) (e) (f) (g) (h) (i) (j) (k) (l) N+
-
Which of the following molecules may be polar? (a) CH3Cl (C3J, (b) HW2 (CO)10 (D4h), (c) SnCl4 (Td).
-
Which of the following molecules may show a pure rotational microwave absorption spectrum? (a) H20, (b) H202, (c) NH4 (d) N20?
-
Narrowly defining public stock companies in terms of financial performance can lead to Blank______. improved public relations more accurate accounting black swan events more efficient production
-
During 2015, Merkley Company disposed of three different assets. On January 1, 2015, prior to their disposal, the accounts reflected the following: The machines were disposed of in the following...
-
You have been hired as a consultant to assist the following companies with cost-volume-profit analysis: Freemans Retail Floral Shop Susquehanna Trails Bus Service Wilson Pump Manufacturers McCauley &...
-
Determine whether each of the following is true or false. In each case, assume \(P\) is located at \(t=0\) and \(F\) is located at \(t=n\), and the \(A\) s are spread uniformly over the planning...
-
Starbucks understands the important role each participant in its value chain plays. Starbucks offers a selection of coffees from around the world. There are many potential challenges in transforming...
-
The following data is provided for Garcon Company and Pepper Company for the year ended December 31. Finished goods inventory, beginning Work in process inventory, beginning Raw materials inventory,...
-
KEW Enterprises began operations in January 2015 to manufacture a hand sanitizer that promised to be more effective and gentler on the skin than existing products. Family members, one of whom was...
-
Located on the Melbourne campus of Edvantage Institute Australia, Edvantage Sport is a fitness and aquatics centre that provide a wide range of facilities and programs to keep their members active...
-
In the 2022/23 income year, Ironclad made three frankable distributions. The first distribution ($1 million) is franked at 50%, the second distribution ($2million) is franked at 70%, and the third...
-
Assuming the role of an analyst, tasked by Kimi Ford with reviewing Joanna Cohen's analysis, you will write a formal executive summary detailing your evaluation. This evaluation should be focused on...
-
Jason Inc. purchases a patent with a legal life of 10 years but due to technology, the patent is expected to not have relevance after 5 years. The total capitalized costs for the patent are $10,000....
-
Silja is a single taxpayer. She earned wages of $7,000 from a part-time job in 2023 but her primary endeavor was her new small business that unfortunately generated an ordinary tax loss of $18,000....
-
A menu items costs need to take the following into consideration: cost of food and beverage, other direct costs (such as plate garnishes and packing used for take out orders), and an allowance for...
-
2. Which would you prefer to receive, if you can invest the funds at a 5% annual return? Find the present value for each option. a) $1,000 a year for 10 years, or b) $800 a year for 15 years at 5%...
-
What are the four types of poultry production systems? Explain each type.
-
Hydrolysis of atropine gives tropine and ()-tropic acid. (a) What are their structures? (b) Even though tropine has a chirality center, it is optically inactive. Explain. (c) An isomeric form of...
-
Acid-catalyzed dehydration of either 2-methyl-1-butanol or 3-methyl-1-butanol gives 2-methyl-2-butene as the major product. Write plausible mechanisms that explain these results.
-
Propose structures for two constitutional isomers of cyclopentene that do not contain a ring. Cyclopentene
-
Use the magnitudes (Richter scale) of the earthquakes listed in Data Set 16 in Appendix B. In 1989, the San Francisco Bay Area was struck with an earthquake that measured 7.0 on the Richter scale....
-
Based on data from Data Set 17 in Appendix B, the words spoken in a day by women have a mean of 16,215 words and a standard deviation of 7301 words. Consider the women with an unusual word count in a...
-
Measures of Location The values of P 50 , Q 2 , and the median are found for the net incomes reported on all individual 1040 tax forms filed last year. What do those values have in common?

Study smarter with the SolutionInn App


