Match each chemical reaction with the correct diagram according to Hess' Law. Enthalpy, , =...
Fantastic news! We've Found the answer you've been seeking!
Question:
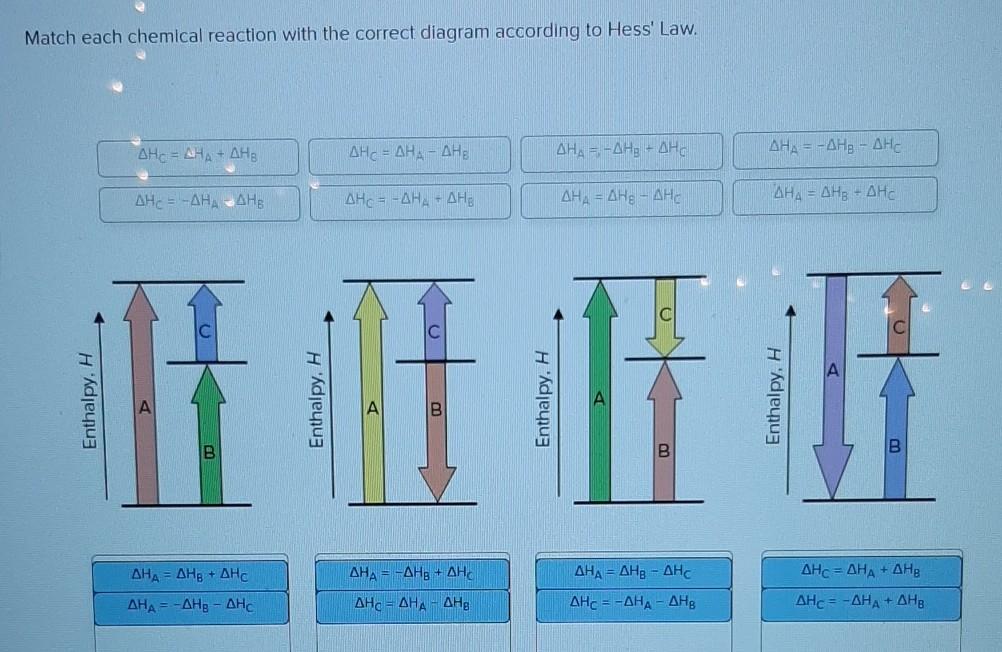
Transcribed Image Text:
Match each chemical reaction with the correct diagram according to Hess' Law. Enthalpy, Η ΔΗ, = ΔΗΑ + ΔΗΒ ΔΗΞ -ΔΗ, ΔΗΒ ΔΗΑ = ΔΗΒ + ΔΗ ΔΗ, = - ΔΗg – ΔΗ Enthalpy. Η 7 ΔΗς = ΔΗΑ – ΔΗ ΔΗΕ = - ΔΗΑ - ΔΗΒ ΔΗ, = −ΔΗg + ΔΗ ΔΗο = ΔΗΑ - ΔΗΒ Enthalpy. Η ΔΗ, = - ΔΗ; - ΔΗ ΔΗ, = ΔΗΕ - ΔΗ B ΔΗ, = ΔΗg - ΔΗ ΔΗc = −ΔΗΑ – ΔΗΒ ΔΗΑ = - ΔΗg - ΔΗ ΔΗ, = ΔΗΒ + ΔΗ Enthalpy. Η Β ΔΗς = ΔΗΑ + ΔΗΒ ΔΗc = - ΔΗΑ + ΔΗΒ Match each chemical reaction with the correct diagram according to Hess' Law. Enthalpy, Η ΔΗ, = ΔΗΑ + ΔΗΒ ΔΗΞ -ΔΗ, ΔΗΒ ΔΗΑ = ΔΗΒ + ΔΗ ΔΗ, = - ΔΗg – ΔΗ Enthalpy. Η 7 ΔΗς = ΔΗΑ – ΔΗ ΔΗΕ = - ΔΗΑ - ΔΗΒ ΔΗ, = −ΔΗg + ΔΗ ΔΗο = ΔΗΑ - ΔΗΒ Enthalpy. Η ΔΗ, = - ΔΗ; - ΔΗ ΔΗ, = ΔΗΕ - ΔΗ B ΔΗ, = ΔΗg - ΔΗ ΔΗc = −ΔΗΑ – ΔΗΒ ΔΗΑ = - ΔΗg - ΔΗ ΔΗ, = ΔΗΒ + ΔΗ Enthalpy. Η Β ΔΗς = ΔΗΑ + ΔΗΒ ΔΗc = - ΔΗΑ + ΔΗΒ
Expert Answer:
Answer rating: 100% (QA)
ACcording to Hesss law ia a reaction tales place in multiple steps then the total enthallpy change o... View the full answer

Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
Determine the equilibrium concentrations for this chemical reaction with the given Keq. CO(g) +H2O(g)+ 0.750 M 0.750 M CO2 (g) +H2(g) K,q=16.0
-
Determine the equilibrium concentrations for this chemical reaction with the given Keq. H,S(g)+ 0.882 M H2 (g) + S(s) K-= 0.055
-
Determine the equilibrium concentrations for this chemical reaction with the given Keq. K.~ 1.2x10 1.99M
-
The passage indicates that the late 1850s Democrats: F. Were all Southern slaveholders who wanted to expand slavery into the territories. G. Used legislation in the early 1850s to support their...
-
The marginal cost, marginal revenue, and demand curves for Houston Home and Gardens deluxe wheelbarrow are shown in the graph on the shown below. Required: Before completing any of the following...
-
Discuss the concept of corporate social responsibility. What role does it play in the relationship between a company and its host country? How does CSV move beyond CSR?
-
A 1.0-m-long, 1.0-mm-diameter copper wire carries a current of 50.0 A to the east. Suppose we create a magnetic field that produces an upward force on the wire exactly equal in magnitude to the...
-
Active Memories Inc. manufactures RAM memory chips for personal computers. An activity analysis was conducted, and the following activity costs were identified with the manufacture and sale of memory...
-
Mickley Company's plantwide predetermined overhead rate is $21.00 per direct labor-hour and its direct labor wage rate is $14.00 per hour. The following information pertains to Job A-500: Direct...
-
An Expert Witness can be anyone with knowledge or experience of a particular field or discipline beyond that to be expected of a layman. The Expert Witness's duty is to give to the Court or tribunal...
-
The Arsen Co. earned $500,000 last year. The company had 100,000 shares outstanding on January 1, sold 6,000 shares on June 1 and sold 9,000 shares on August 1. The Arsen Co. stock sells for $50 per...
-
What is organizational culture? Explain the functions for organizational culture.
-
Why is organizational behavior important to a company? ( Perhaps focus on "Organizational Effectiveness", profit... )
-
What is the Evolution and Implementation of Business Ethics and Corporate Social Responsibility?
-
Discuss the different approaches to diversity training. What do you see as the pro's and con's of each approach? Explain
-
Describe four approaches to quality management. Which approach do you believe is most effective? Why?
-
Alpha Beta Co. (ABC) is trying to determine the initial outlay of a project. ABC estimates that the project will generate a $25,000 net cash flow at the end of each year for 20 years. The project's...
-
A firm offers two products for sale. The marginal cost of one product is new zero once the first unit has been produced. The marginal cost of the other product rises as output rises. What would be...
-
A flask contains a 30.0 mL sample of acetone (nail polish remover) that weighs 23.6 grams. What is the density of the acetone?
-
A ruby laser puts out a pulse of red light at a wavelength of 694 nm. (The ruby laser is described in the essay on p. 226.) If a pulse delivers 1.05 10 5 watts of power for 280 s, how many photons...
-
What hybrid orbitals would be expected for the central atom in each of the following molecules or ions? a. SeI 2 b. NO 3 - c. BeCl 2 d. ClO 4 -
-
Find the position of the first minimum for a single slit of width 0.04 \(\mathrm{mm}\) on a screen of \(2 \mathrm{~m}\) distance, when light from a He-Ne laser \(\lambda=\) 6328 is shone on the slit.
-
A diffraction grating is a closely spaced array of apertures or obstacles forming a series of closely spaced slits. The simplest type in which an incoming wave front meets alternating opaque and...
-
(a) Determine the coherence area for a mercury arc lamp at 6330 at a distance of \(1 \mathrm{~m}\) from the source. Assume that the output aperture is \(3 \mathrm{~mm}\) and that the beam is...

Study smarter with the SolutionInn App


