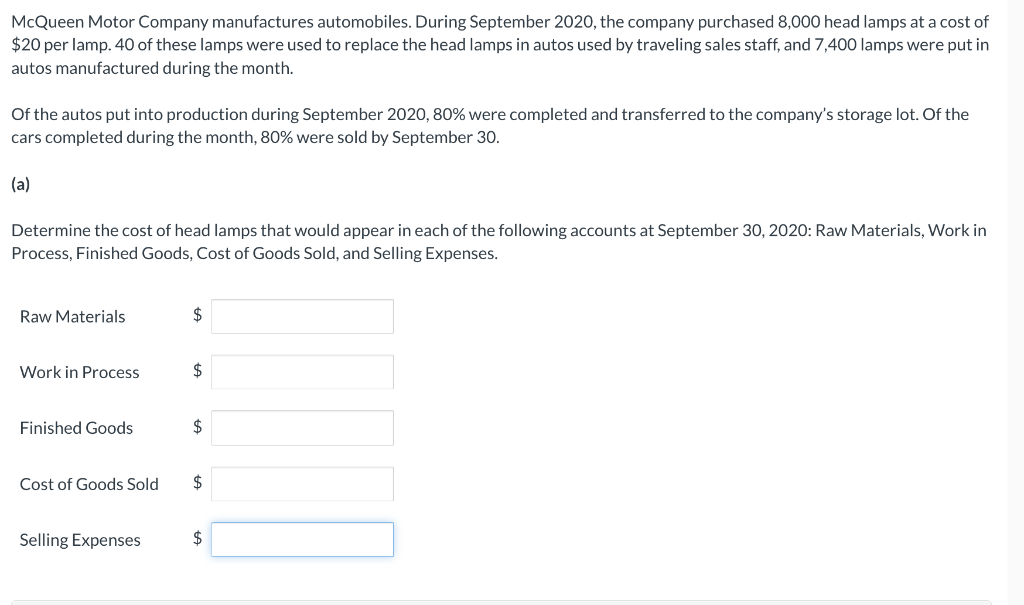
McQueen Motor Company manufactures automobiles. During September 2020, the company purchased 8,000 head lamps at a...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
McQueen Motor Company manufactures automobiles. During September 2020, the company purchased 8,000 head lamps at a cost of $20 per lamp. 40 of these lamps were used to replace the head lamps in autos used by traveling sales staff, and 7,400 lamps were put in autos manufactured during the month. Of the autos put into production during September 2020, 80% were completed and transferred to the company's storage lot. Of the cars completed during the month, 80% were sold by September 30. (a) Determine the cost of head lamps that would appear in each of the following accounts at September 30, 2020: Raw Materials, Work in Process, Finished Goods, Cost of Goods Sold, and Selling Expenses. Raw Materials $ Work in Process $ Finished Goods $ Cost of Goods Sold $ Selling Expenses $ McQueen Motor Company manufactures automobiles. During September 2020, the company purchased 8,000 head lamps at a cost of $20 per lamp. 40 of these lamps were used to replace the head lamps in autos used by traveling sales staff, and 7,400 lamps were put in autos manufactured during the month. Of the autos put into production during September 2020, 80% were completed and transferred to the company's storage lot. Of the cars completed during the month, 80% were sold by September 30. (a) Determine the cost of head lamps that would appear in each of the following accounts at September 30, 2020: Raw Materials, Work in Process, Finished Goods, Cost of Goods Sold, and Selling Expenses. Raw Materials $ Work in Process $ Finished Goods $ Cost of Goods Sold $ Selling Expenses $
Expert Answer:
Answer rating: 100% (QA)
Purchases 8000 20 40 used in sales staff travelling autos 7400 used in produ... View the full answer

Related Book For 

Accounting Principles
ISBN: 978-1119411482
13th edition
Authors: Jerry J. Weygandt, Paul D. Kimmel, Donald E. Kieso
Posted Date:
Students also viewed these accounting questions
-
A company fabricates a component at a cost of $6.00. A supplier offers to supply the same component for $5.50. Under what circumstances is it reasonable to purchase from the supplier?
-
A company fabricates a company at a cost of $6.00. A supplier offers to supply the same component for $5.50. Under what circumstances is it reasonable to purchase from the supplier?
-
A company fabricates a component at a cost of $4.00. A supplier offers to supply the same component for $3.60. Under what circumstances is it reasonable to purchase from the supplier?
-
Calculate e y for the following values of y: (a) y = 1 (b) y = 5 (c) y = -.5 (d) y = -2.5 (e) y = 3.1 (f) y = - I (g) y = .05 (h) y = .32 (i) y = 6.1 (j) y = -5.4
-
Bell Tolls, Inc., has an average collection period of 36 days. Its average daily investment in receivables is $58,300. What are annual credit sales? What is the receivables turnover?
-
In Problem convert the given interest rate to decimal form if it is given as a percentage, and to a percentage if it is given in decimal form. 0.0084
-
When engaged to express an opinion on one or more specified elements, accounts, or items of a financial statement, an auditor: a. May not describe auditing procedures applied. b. Should advise that...
-
Wu Company's income statement for the year ended December 31, 2011, and its comparative balance sheets as of December 31, 2011 and 2010, are presented on the next page. During 2011, Wu Company...
-
San Diego Corp has 2,318,809 million shares of common stock outstanding witha par value of $1.00 per share. The current marketprice per share is $ 75. San Diego Corp hasoutstanding debt with a par...
-
Suppose the smartphone manufacturer Peony Electronics provides the following information for its costs last month (in millions): Suppose the smartphone manufacturer Peony Electronics provides the...
-
Walton Manufacturing Company gathered the following data for the month. Cost of goods sold: $35,000; Sales: $89,000; selling expenses: $16,000; Administrative expenses: $21,000. How much net...
-
(Apply legal principles in property law matters) Imagine you are working as a conveyancing clerk. Your newly appointed supervisor comes to you and says that he is revising a property sale and seems...
-
Analyse the Land and Business (Sale and Conveyancing) Act 1994 that applies to South Australia. report that includes: 4003RE02Q02: Explain the purpose of the Land and Business (Sale and Conveyancing)...
-
Selected accounts with amounts omitted are as follows Work in Process Aug. 1 Balance 2 5 4 , 0 8 0 Aug. 3 1 Goods finished 1 2 3 , 9 2 0 3 1 Direct materials X 3 1 Direct labor 3 7 , 0 0 0 3 1...
-
Big Data, EHR & Healthcare Professionals Legal & Ethical Issues INSTRUCTIONS Copy the questions for Parts I - II below into a Word document. Part I: The Articles a. Electronic Health Records: ( 1)...
-
SCENARIO: Little Tummy Foods (LTF) is an Auckland-based company that manufactures "on-the-go" snacks, such as muffins, nut mixes, and muesli bars. Over the past years, the brand has gained...
-
The Great Falls Cheese Company wants to determine how well it estimated its Direct Material costs by comparing the standards it predicted to actual data. Great Falls has the following standards:...
-
The Thomas Corporation was organized on Jan. 1, 2020. On Dec. 31, 2021, the corporation lost most of its inventory in a warehouse fire before the year-end count of inventory was to take place. just...
-
Dieker Company begins operations on January 1. Because all work is done to customer specifications, the company decides to use a job order cost system. Prepare a flowchart of a typical job order...
-
Hawke Company had the following assets and liabilities on the dates indicated. Hawke began business on January 1, 2019, with an investment of $100,000. Instructions From an analysis of the change in...
-
Who are the two key international players in the development of international accounting standards? Explain their role.
-
(a) Describe the effect of temperature on the equilibrium constant. (b) Industrial \(\mathrm{CH}_{3} \mathrm{OH}\) is prepared according to the reaction \[ \mathrm{CO}(\mathrm{g})+2...
-
Under atmospheric conditions the acetone-chloroform azeotrope boils at \(64.6^{\circ} \mathrm{C}\) and contains 33.5 mole per cent acetone. The vapour pressures of acetone and chloroform at this...
-
Explain in detail, with neat sketch, the working principle of a fuel cell in producing electrical energy from the chemical energy of a fuel.

Study smarter with the SolutionInn App


