A. Solubility of Solids in Water and Cyclohexane Fill in the table with your solubility data...
Fantastic news! We've Found the answer you've been seeking!
Question:
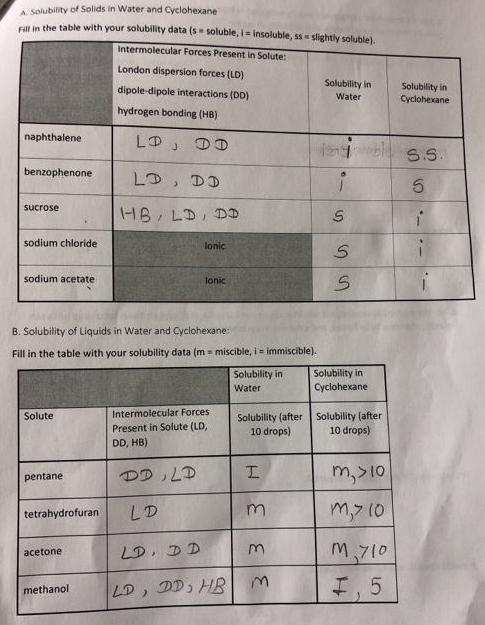


Transcribed Image Text:
A. Solubility of Solids in Water and Cyclohexane Fill in the table with your solubility data (s soluble, i-insoluble, ss slightly soluble). Intermolecular Forces Present in Solute: naphthalene benzophenone sucrose sodium chloride sodium acetate Solute pentane tetrahydrofuran B. Solubility of Liquids in Water and Cyclohexane: Fill in the table with your solubility data (m= miscible, i = immiscible). acetone London dispersion forces (LD) dipole-dipole interactions (DD) hydrogen bonding (HB) DD methanol LD, DD HB, LD, DD lonic Tonic Intermolecular Forces Present in Solute (LD, DD, HB) DD, LD LD LD, DD LD, DD, HB Solubility in Water Solubility (after 10 drops) H 33 3 Solubility in Water S SS Solubility in Cyclohexane Solubility (after 10 drops) m, >10 m,> 10 M710 I. 5 Solubility in Cyclohexane S.S. 5 4. For the molecular compounds you investigated in Parts A and B, explain the relationship between polarity, intermolecular forces, and solubility in water. Provide specific examples from part A: Provide specific examples from part B: 5. For the molecular compounds you investigated in Parts A and B, explain the relationship between polarity, intermolecular forces, and solubility in cyclohexane. Provide specific examples from part A: Provide specific examples from part B: 10 6. Are the ionic compounds you investigated in Part A more soluble in water or cyclohexane? What is the reason for this observed behavior? A. Solubility of Solids in Water and Cyclohexane Fill in the table with your solubility data (s soluble, i-insoluble, ss slightly soluble). Intermolecular Forces Present in Solute: naphthalene benzophenone sucrose sodium chloride sodium acetate Solute pentane tetrahydrofuran B. Solubility of Liquids in Water and Cyclohexane: Fill in the table with your solubility data (m= miscible, i = immiscible). acetone London dispersion forces (LD) dipole-dipole interactions (DD) hydrogen bonding (HB) DD methanol LD, DD HB, LD, DD lonic Tonic Intermolecular Forces Present in Solute (LD, DD, HB) DD, LD LD LD, DD LD, DD, HB Solubility in Water Solubility (after 10 drops) H 33 3 Solubility in Water S SS Solubility in Cyclohexane Solubility (after 10 drops) m, >10 m,> 10 M710 I. 5 Solubility in Cyclohexane S.S. 5 4. For the molecular compounds you investigated in Parts A and B, explain the relationship between polarity, intermolecular forces, and solubility in water. Provide specific examples from part A: Provide specific examples from part B: 5. For the molecular compounds you investigated in Parts A and B, explain the relationship between polarity, intermolecular forces, and solubility in cyclohexane. Provide specific examples from part A: Provide specific examples from part B: 10 6. Are the ionic compounds you investigated in Part A more soluble in water or cyclohexane? What is the reason for this observed behavior?
Expert Answer:
Answer rating: 100% (QA)
ATTACHED IMAGE Answer 4 As the polarity of the solvent increases its solubility also increases Polar ... View the full answer

Related Book For 

Chemistry The Central Science
ISBN: 978-0321696724
12th edition
Authors: Theodore Brown, Eugene LeMay, Bruce Bursten, Catherine Murphy, Patrick Woodward
Posted Date:
Students also viewed these accounting questions
-
Water and diethyl ether are immiscible liquids. Charged compounds dissolve in water, and uncharged compounds dissolve in ether. a. What pH would you make the water layer in order to cause both...
-
In general, the attractive intermolecular forces between solvent and solute particles must be comparable or greater than solute-solute interactions for significant solubility to occur. Explain this...
-
The following compounds are only slightly soluble in water, but one of them is very soluble in a dilute aqueous solution of sodium hydroxide. The other is still only slightly soluble. (a) Explain...
-
Its estimated that the average corporate user sends and receives some 112 e-mails daily.78 Thats about 14 e-mails per hour, and even if half of those dont require a lot of time and concentration,...
-
Buford sells an apartment building for $720,000. His adjusted basis is $500,000 for regular income tax purposes and $550,000 for AMT purposes. Calculate Buford's: a. Gain for regular income tax...
-
As stated in the chapter, most accounting-related errors are detected and corrected in the current period. Of those that go undetected, some will fix themselves over two periods, while other errors...
-
Do insurance companies use race as a determining factor when making insurance available? Fienberg (1985) gathered data from a report issued by the U.S. Commission on Civil Rights about the number of...
-
On January 1, 2012, Zakiuddin Company purchased the following two machines for use in its production process. Machine A: The cash price of this machine was $55,000. Related expenditures included:...
-
The following cost and inventory data are taken from the accounting records of Mason Company for the year just completed: Costs incurred: Direct labour cost Purchases of raw materials. Indirect...
-
Following are the ordinates of a storm hydrograph of a river draining a catchment area of 423 km due to a 6h storm.. Derive the ordinates of 6h unit hydrograph & plot the same. Time in hr -6 Stream...
-
Year over year, online shopping has increased its market share, but what impact has COVID-19 had on e-commerce? And what, if any, impact has the rise of online shopping had on the supply chain? Are...
-
You borrow $25,000 for a home repair. You can afford to pay back $800 a month. If you borrow the money directly from a bank, you have a 6.25% interest rate. How longwill it take you to pay off the...
-
Suppose that an investor has a 6-year investment horizon. The investor is considering a 13-year 9% coupon bond selling at its par value of $1,000. The investor's expectations are as follows: 1. The...
-
Grouper Corporation's adjusted trial balance contained the following asset accounts at December 31, 2023: Prepaid Rent $22,900; Goodwill $52,000; Franchise Fees Receivable $3,000; Intangible...
-
A $1,000 face value bond has a 5% coupon rate and matures after eight years. The bond is currently selling for $850. What is the yield to maturity on this bond? I
-
we are assuming 1 share of stock for 1 Put Option. An investor buys a Put Option of Widget stock on March 1. Option Price: $6.00/1 share of Stock; Expiration Date June 1; Strike Price: $140; Current...
-
An economy is believed to have a marginal propensity to tax of 0.25, a marginal propensity to import of 0.2, and a multiplier value of 1.25. The value of the marginal propensity to consume is...
-
A horizontal annulus with inside and outside diameters of 8 and 10 cm, respectively, contains liquid water. The inside and outside surfaces are maintained at 40 and 20oC, respectively. Calculate the...
-
The following is known as the thermite reaction [Figure 5.8(b)]: This highly exothermic reaction is used for welding massive units, such as propellers for large ships. Using standard enthalpies of...
-
Does the following drawing represent a neutral atom or an ion? Write its complete chemical symbol including mass number, atomic number, and net charge (if any). 16 protons+ 16 neutrons 18 electrons
-
Name or write the condensed structural formula for the following compounds: (a) Trans -2-pentene (b) 2,5-dimethyl-4-octene CH3 (c) CH3CH2 CH2CHCH2CH3 - (d) Br Br CH2CH3 CH3
-
The trial balance of Hindustan Textiles Ltd. as at 31st March 2006 is as presented hereunder. Further information 1.The authorized capital of the company is 30 lakh equity shares of 10 each of which...
-
Refer to the exercise on preparation and brief analysis of financial statements of Sangeeta Udhuog Ltd. as given in the last chapter. Now convert the financial statements in to vertical format. Take...
-
Gupta Health Products Pvt. Ltd. has been promoted by Richa Gupta and her associates on April 1, 20CY. The company enters into the following transactions of purchase during the month. It maintains a...

Study smarter with the SolutionInn App


