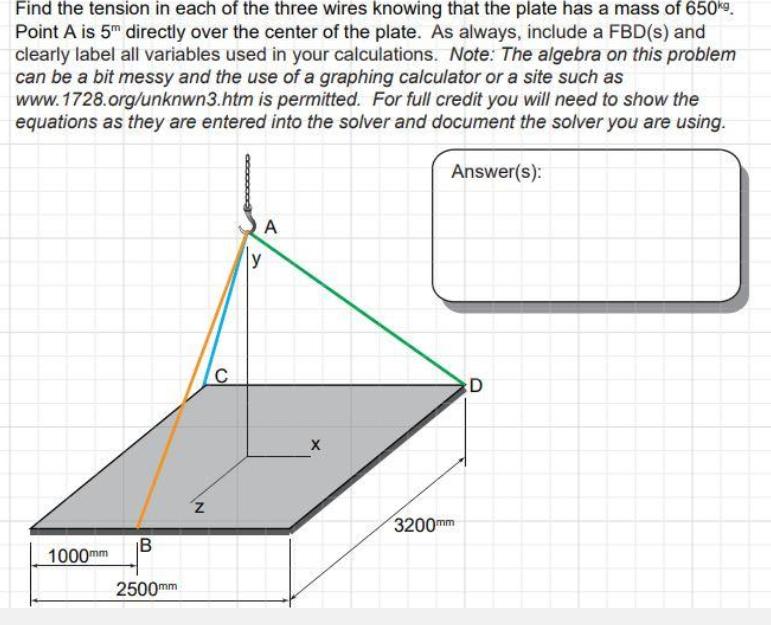
Find the tension in each of the three wires knowing that the plate has a mass...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
Find the tension in each of the three wires knowing that the plate has a mass of 650kg. Point A is 5" directly over the center of the plate. As always, include a FBD(s) and clearly label all variables used in your calculations. Note: The algebra on this problem can be a bit messy and the use of a graphing calculator or a site such as www.1728.org/unknwn3.htm is permitted. For full credit you will need to show the equations as they are entered into the solver and document the solver you are using. Answer(s): A y C D 3200mm B 1000mm 2500mm Find the tension in each of the three wires knowing that the plate has a mass of 650kg. Point A is 5" directly over the center of the plate. As always, include a FBD(s) and clearly label all variables used in your calculations. Note: The algebra on this problem can be a bit messy and the use of a graphing calculator or a site such as www.1728.org/unknwn3.htm is permitted. For full credit you will need to show the equations as they are entered into the solver and document the solver you are using. Answer(s): A y C D 3200mm B 1000mm 2500mm
Expert Answer:

Related Book For 

Physics
ISBN: 978-0077339685
2nd edition
Authors: Alan Giambattista, Betty Richardson, Robert Richardson
Posted Date:
Students also viewed these civil engineering questions
-
Find the tension in each cord in Fig. 5.44 if the weight of the suspended object is w. 45 60// ic
-
Find the tension in each cable inFigure 30.0 45.0 T. T. 3 475 lb
-
Find the tension in each cable inFigure. 50.0 T. T. 3 2200 N
-
Consider a long cylindrical solenoid with diameter R, number of current loops N and length L through which a current I runs. Now (a) Use Ampre's law to calculate the magnetic field inside the...
-
Name the following molecules according to the IUPAC system of nomenclature. (a) (b) (c) (d) (e) CH3CH(CH3)CH(CH3)CH(CH3)CH(CH3)2 (f) CH3CH2CHCH3 CH CH H3C CH3 CHCHCH2CH3 EH CHCH,CH.CCH,CH.CH CH,...
-
The beam is used to support a dead load of 400 lb/ft, a live load of 2 k/ft, and a concentrated live load of 8 k. Determine (a) The maximum positive vertical reaction at A, (b) The maximum positive...
-
Does Fairmont have any contactor personnel whose have terminated but are being paid through payroll after termination (e.g., ghost employees)?
-
The citizens of Spencer County approved the issuance of $2,000,000 in 6 percent general obligation bonds to finance the construction of a courthouse annex. A capital projects fund was established for...
-
A 20 kg dog requires treatment with ketoconazole at 5 mg/kg by oral suspension. The suspension contains 100 mg/5 mL oral solution. What volume does the dog require per dose?
-
Darden Restaurants, Inc. (DRI) is the largest full-service restaurant company in the world. It operates over 2,200 restaurants under a variety of brand names, including Olive Garden, Bahama Breeze,...
-
Another famous example of linear relationships is relating specific lengths of bones in skeletal remains to the height of a person, so that anthropologists can estimate the height of a person when...
-
@M zain do not respond to this question again!!! You need to stop posting ridiculous solutions to my question. You literally respond in seconds after this question that I post with crazy weird...
-
Sunland Sdn. Bhd. has land, buildings, and machinery as its Plant, Property, and Equipment as of 31 December 2015. The company uses the straight-line depreciation method for all depreciable assets...
-
1.2 Discuss the various modes of delivery (contract of sale) (10 marks) 1.3 Differentiate between suspensive and resolutive condition (5 marks)
-
1.What are the four basic assumptions underlying GAAP? 2.Briefly define the financial accounting elements: (1) assets, (2) liabilities, (3) equity, (4) investments by owners, (5) distributions to...
-
Alka-Seltzer Inc. has decided to purchase some new equipment. Alka-Sletzer can only pay $10,000 of the $200,000 purchase price and must finance the remainder on a long term note. The credit will go...
-
Iron Chemicals acquires a machine that should go through a major overhaul every three years. The total price for the equipment is 1 million. It is estimated that each overhaul will cost 200,000. The...
-
In what ways does a well-designed enterprise search software vary from popular search engines (e.g., Bing, DuckDuckGo, and Google)?
-
A car moving at 30 mi/h is stopped by jamming on the brakes and locking the wheels. The car skids 50 ft before coming to rest. How far would the car skid if it were initially moving at 60 mi/h? [You...
-
A water sample is found to have 0.016% deuterium content (that is, 0.016% of the hydrogen nuclei in the water are 2H). If the fusion reaction (2H + 2H) yields 3.65 MeV of energy on average, how much...
-
2.0 102 m, (a) What is the thermal resistance if the material is asbestos? (b) What is the thermal resistance if the material is iron? (c) What is the thermal resistance if the material is copper?
-
How does each of the following variables behave over the business cycle? Develop graphs to show your results and give economic explanations. a. Real imports b. Federal government receipts c. Housing...
-
In the FRED database, find a variable that is available in both a seasonally adjusted form and a not seasonally adjusted form. Plot both over time and describe how large the seasonal variation in the...
-
It has been argued that the stock market predicts recessions. Using quarterly data since 1961, plot the real value of the stock market index (the Wilshire 5000 index in the last month of the quarter...

Study smarter with the SolutionInn App


