Question: comparing structures to which one is the best and worst guest compund and include why basing it off structure, molecular forces, polarities. thank you
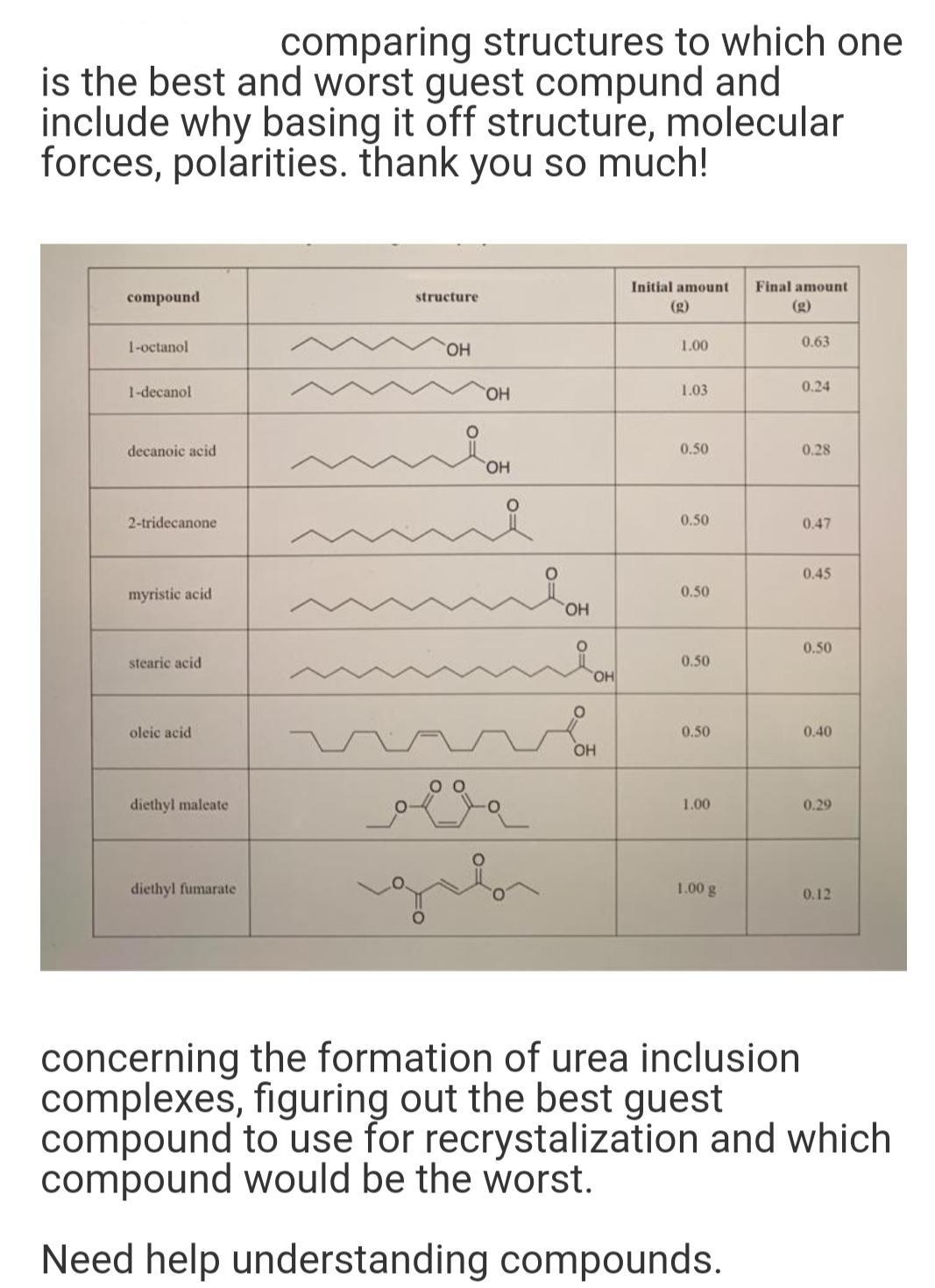
comparing structures to which one is the best and worst guest compund and include why basing it off structure, molecular forces, polarities. thank you so much! Initial amount Final amount compound structure (g) (g) 1-octanol HO. 1.00 0.63 1-decanol HO. 1.03 0.24 decanoic acid 0.50 0.28 HO. 2-tridecanone 0.50 0.47 0.45 myristic acid 0.50 HO. 0.50 stearic acid 0.50 HO, oleic acid 0.50 0.40 OH diethyl maleate 1.00 0.29 diethyl fumarate 1.00 g 0.12 concerning the formation of urea inclusion complexes, figuring out the best guest compound to use for recrystalization and which compound would be the worst. Need help understanding compounds.
Step by Step Solution
3.40 Rating (150 Votes )
There are 3 Steps involved in it
Here is the explained answer Step 1 Molecular forces An intermolecular force IMF or secondary force ... View full answer

Get step-by-step solutions from verified subject matter experts


