Question: Test each listed substance with the five indicators. Determine the pH for the substance and record. red litmus (acid/base?) (acid/base?) (Hydrion) blue litmus phenolphthalein
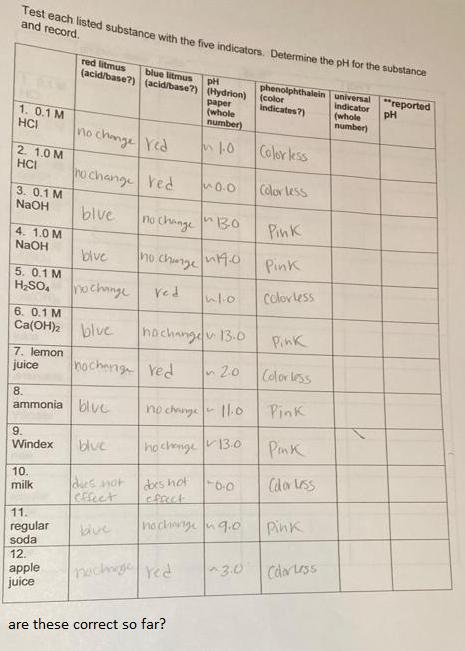
Test each listed substance with the five indicators. Determine the pH for the substance and record. red litmus (acid/base?) (acid/base?) (Hydrion) blue litmus phenolphthalein universal "reported (color indicates?) pH paper (whole number) Indicator (whole number) pH 1. 0.1 M HCI change Yed no Color ess 2. 1.0 M HCI no change red 0.0 Color tess 3. 0.1 M NaOH blve no change 30 Pink 4. 1.0 M NaOH blve no.chunge Pink 5. 0.1 M H,SO, nochange Ved Colovless 6. 0.1 M Ca(OH) blve hochangdv 13-0 Pink 7. lemon juice nocheng ved n20 Colorless 8. ammonia blve no chunge Pink 9. Windex blue hochige 13:o Pink 10. milk dbes hot fect Cdoruss des not Cffect 11. no chwnge uq.0 Pink regular soda 12. 3.0 Cdar Less apple pachg red juice are these correct so far?
Step by Step Solution
3.36 Rating (177 Votes )
There are 3 Steps involved in it
The ans... View full answer

Get step-by-step solutions from verified subject matter experts
Document Format (2 attachments)
6360d37587d0b_233036.pdf
180 KBs PDF File
6360d37587d0b_233036.docx
120 KBs Word File


