Needed Information Molar mass of Ba(OH)2 = 171.35 g/mol If 50.0 grams of Ba(OH), is...
Fantastic news! We've Found the answer you've been seeking!
Question:
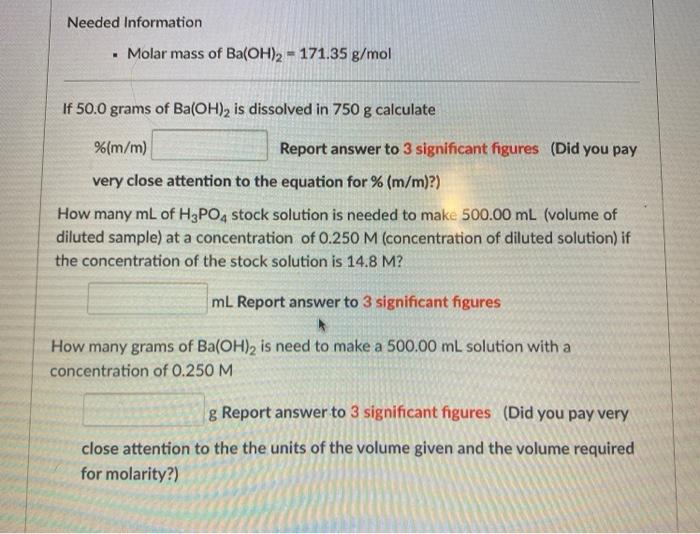
Transcribed Image Text:
Needed Information • Molar mass of Ba(OH)2 = 171.35 g/mol If 50.0 grams of Ba(OH), is dissolved in 750 g calculate %(m/m) Report answer to 3 significant figures (Did you pay very close attention to the equation for % (m/m)?) How many mL of H3PO, stock solution is needed to make 500.00 mL (volume of diluted sample) at a concentration of 0.250 M (concentration of diluted solution) if the concentration of the stock solution is 14.8 M? mL Report answer to 3 significant figures How many grams of Ba(OH)2 is need to make a 500.00 mL solution with a concentration of 0.250 M g Report answer to 3 significant figures (Did you pay very close attention to the the units of the volume given and the volume required for molarity?) Needed Information • Molar mass of Ba(OH)2 = 171.35 g/mol If 50.0 grams of Ba(OH), is dissolved in 750 g calculate %(m/m) Report answer to 3 significant figures (Did you pay very close attention to the equation for % (m/m)?) How many mL of H3PO, stock solution is needed to make 500.00 mL (volume of diluted sample) at a concentration of 0.250 M (concentration of diluted solution) if the concentration of the stock solution is 14.8 M? mL Report answer to 3 significant figures How many grams of Ba(OH)2 is need to make a 500.00 mL solution with a concentration of 0.250 M g Report answer to 3 significant figures (Did you pay very close attention to the the units of the volume given and the volume required for molarity?)
Expert Answer:
Related Book For 

Fundamentals of Analytical Chemistry
ISBN: 978-0495558286
9th edition
Authors: Douglas A. Skoog, Donald M. West, F. James Holler, Stanley R. Crouch
Posted Date:
Students also viewed these accounting questions
-
A thin, 50.0-cm-Iong metal bar with mass 750 g rests n, but is not attached to, two metallic supports in a uniform 0.450-Tmagnetic field, as shown v R in Fig. A battery and a 25.0-Ω resistor in...
-
Calculate the mass in grams of the following. a. 0.15 mol Na b. 0.594 mol S c. 2.78 mol CH2Cl2 d. 38 mol (NH4)2S
-
Calculate the mass in grams of the following. a. 0.205 mol Fe b. 0.79 mol F c. 5.8 mol CO2 d. 48.1 mol K2CrO4
-
Two tiny particles having charges of 5.76 C and -7 C are placed along the y-axis. The 5.76 C particle is at y = 0 cm, and the other particle is at y = 48.74 cm. Where must a third charged particle be...
-
Examine High Liner's statements of cash flows. Explain and interpret the information in the statement and discuss what it tells you about High Liner.
-
The following results were obtained from an experiment carried out to measure the enthalpy of combustion (heat of combustion) of ethanol. The experiment involved heating a known volume of water with...
-
Use the equation (17.19) suggested by Pai for the turbulent stress and integrate for the velocity profile. How do the results compare with that of Prandtl? = 0.9835 H+ (1-#)['-(1-#)'] (17.19) H+
-
Auburn Concrete Inc. is considering the purchase of a new concrete mixer to replace an inefficient older model. If purchased, the new machine will cost $90,000 and is expected to generate savings of...
-
Write a Java record (Class) Car with the following features (Instance variables): Make, Model, Color, and Year. Declare an array Cars to store 10 cars. Define an instance method setCars() to populate...
-
A national catalog and Internet retailer has three warehouses and three major distribution centers located around the country. Normally, items are shipped directly from the warehouses to the...
-
After each department's network is allocated its IP addresses, how many addresses from the initial block 130.216.4.0/23 will remain unallocated? How do I find the number of addresses in that initial...
-
P21.1B (LO 2,4) (Lessee Entries, Finance Lease) The following facts pertain to a non-cancelable lease agreement between Jupiter Leasing Company and Siskiwit Company, a lessee: Commencement date...
-
Research on quality standards associated with residential construction to determine applicable standards.quality objectives associated with the project should be defined. 1. quality standards that...
-
What is the internal rate of return per period for a project that requires an investment of 25,000 and returns 5,478 for 7 periods?
-
Managing Multicultural Team (due Week Five, Sunday, November 20, 2022): Describe how people and organizations work locally and internationally - while embracing the multicultural nature of global...
-
A logistics company has hired a new operator to work in the warehouse, in activities related to the reception and dispatch of merchandise and its correct location during the time it remains in the...
-
Which of the following should be used to manage outside cables that need to be routed to various multimode uplinks? Fiber distribution panel 110 punchdown block O A. OB. O C. O D. SOE. PDU...
-
What are the principal differences among asset liquidity management, liability management, and balanced liquidity management?
-
Find the number of half-lives required to reach the following levels of completion: (a) 10%. (b) 90%. (c) 99.9%.
-
Consider the following sets of replicate measurements: Calculate the mean and the standard deviation for each of these six data sets. Calculate the 95% confidence interval for each set of data. What...
-
Calculate the initial potential needed for a current of 0.065 A in the cell Co|Co2+ (5.90 ( 10-3 M) ||Zn2+ (2.95 3 10-3 M) |Zn if this cell has a resistance of 4.50 V.
-
For the data set below, use a partial \(F\)-test to determine whether the variables \(x_{4}\) and \(x_{5}\) do not significantly help to predict the response variable, \(y\). Use the \(\alpha=0.10\)...
-
Suppose we record the gender of the 15 students enrolled in an introductory statistics course as they enter the classroom. The males are denoted by a blue M and the females are denoted by a red...
-
A nutritionist wants to develop a model that describes the relation between the calories, total fat content, protein, sugar, and carbohydrates in cheeseburgers at fast-food restaurants. She obtains...

Study smarter with the SolutionInn App



