Question: Nitrogen from a gaseous phase is to be diffused into pure iron at 700C. If the surface concentration is maintained at the level listed

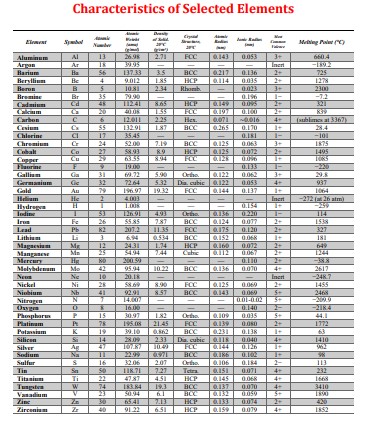


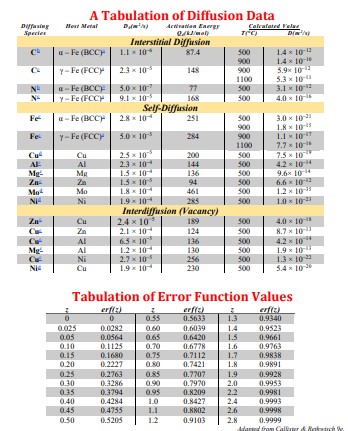
Nitrogen from a gaseous phase is to be diffused into pure iron at 700C. If the surface concentration is maintained at the level listed above, what will be the concentration (in weight percent) with the given case and heat treatment time? The diffusion coefficient for nitrogen in iron at 700C is given above. (Round your answer to the ten thousandths place, 0.0000) Your Answer = Problem 2: For a steel alloy it has been determined that a carburizing heat treatment for the time, given above, will raise the carbon concentration to 0.42 wt% to the depth given from the surface. Estimate the time necessary to achieve the same concentration at the new position stated above for an identical steel and at the same carburizing temperature (in hours). Your Answer Problem 3: The activation energy for the diffusion of species A in metal B is listed above. Calculate the diffusion coefficient at the temperature listed, given that the value of D at 1110 C is 6.2 x 10-11 m/s. Write your answer in scientific notation. Your Answer Characteristics of Selected Elements Element Symbol Number Melting Point("C) Aluminum Al 13 26.98 2.71 FCC 0.143 0.053 3+ 660.4 Argan Ar 18 39.95 Inert -189.2 Barium Ba 56 137.33 3.5 BCC 0.217 0.136 2+ 725 Beryllium Be 4 9.012 185 HCP 0114 0.035 2+ 1278 Berun B 5 10.81 2.34 Rhomb 0.023 3+ 2300 Bromine Br 35 79.90 - 0.196 1- -7.2 Cadmium Cd 48 112.41 8.65 HCP 0.149 0095 2+ 321 20 40.08 1.55 FCC 6.197 0.100 2+ K39 Carbon C 6 12.011 2.25 Hes 0.071 -0.016 4+ (sublimes at 3367) Cesium Cx 55 132.91 1.87 BCC 0.265 0.170 + 28.4 Chlorine 17 35.45 0.181 1- 101- Chromium C 24 52.00 7.19 BCC 0.125 0.063 3+ 1875 Cobalt Co 27 58.93 8.9 HCP 0.125 0.072 2+ 1495 Copper Cu 29 63.35 8.94 FCC 0.128 0.096 1+ 1085 Flaurine F 9 19.00 0.133 -2930 Galliam Gia 31 69.72 5.90 Ortho 0.122 0.062 3+ 29.8 Germanium Ce 32 72.64 5.32 Dia. cubic 0.122 0.053 4+ 937 Gold 79 196.97 19.32 FCC 0.144 0.137 1064 He 2 4.003 - Incr -272 (26 am) Hydrogen H 53 126.91 - 4.93 1+ -259 Ortho 0.136 0.220 T- 114 Fe 26 55.85 7.87 BCC 0.124 0.077 2+ 1538 Lead Pb 82 207.2 11.35 FCC 0.175 0.120 2+ 327 Lithium 3 6.94 0.534 BCC 0.152 0.068 181 Magnesium Mr 12 24.31 1.74 HCP 0.160 0.072 2+ 649 Manganese Ma 25 54.94 2.44 Cubic 6.112 0.067 2+ 1244 Mercury He 80 200.59 0.110 2+ -38.8 Molybdenum Mo 42 95.94 10.22 BCC 0.136 0.070 4+ 2617 Ne 10 20.18 Inert -248.7 Nickel Ni 28 58.69 8.90 FCC 0.125 0.069 2+ 1455 Niobium Nb 41 92.91 8.57 BCC 0.143 0.069 5+ 2468 Nitrogen N T 14.007 - - 0.01-0.02 5+ -2009 Oxygen 0 16.00 - 0.140 2- -2184 Phosphorus P 15 30.97 182 Ortho 0.109 0.035 5+ 44.1 Platinam P 78 195.08 21.45 FCC 0.139 0.060 2+ 1772 Potassium K 19 39.10 0.862 BCC 0.231 0.138 1+ 63 Silicon Si 14 28.09 2.33 Dia cubic 0.118 0.040 4+ 1410 Silver A 47 107.87 10.49 FCC 0.144 0.126 1+ 962 Sodium Na 11 22.99 0971 BCC 0.186 0.102 1+ 98 Sulfur S 16 32.06 207 Ortho. 0.106 0.184 2- 113 Tin Sa 50 118.71 7.27 Tetra 0.151 0.071 4+ 232 Titanium Ti 22 47.87 451 HCP 0.145 0.068 Tungsten W 34 183.84 19.3 BCC 0.137 0.070 4+ 4+ 1668 3410 Vanadium V 23 50.94 6T BCC 0.132 0.059 5+ 1890 30 65.41 7.13 HCP 0.133 0.074 2+ 420 Zirconium 40 91.22 6.51 HCP 0.159 0.079 4+ 1852 Problem: Utilize the Tabulation Error Function Value Table from your Examination Booklet. --Given Values-- Concentration of Nitrogen (wt%): = 0.26 Position in mm = 4.6 Diffusion Coefficient for Nitrogen in Iron at 700C = 6.40E-10 Nitrogen in Iron Heat Treatment Time (hours): = 20 Carburizing Time (hrs): = 7 Steel Alloy Case Depth (mm): 5.6 Steel Alloy New Case Depth (mm) = 8.7 Activation Energy kJ/mol: = 106 Temperature for Problem 3 (Celsius): = 837 -= Data for Problem 4 Temperature in C: = 599 Diffusivity (m/s) = 5.48E-14 Temperature in C: = 697 Diffusivity (m/s) = 3.68E-13 Problem 4: Part A The diffusion coefficients for carbon in nickel are given at two temperatures above. Find the value of Do (m/s): Your Answer Problem 4: Part B The diffusion coefficients for carbon in nickel are given at two temperatures above. Find the value of Qd (kJ/mol): Your Answer: Problem 4: Part C What is the diffusivity, D, at 820 C? Your Answer A Tabulation of Diffusion Data Diffasing Species Host Metal D) Activation Energy Q/kJ/mol) Calculated Value TC) D(x) Interstitial Diffusion a-Fe (BCC) 1.1 10 87.4 500 1.4 10-12 900 1.4 10" Y-Fe (FCC) 2.3 10 148 900 5.9 10-12 1100 5.3 x 10-1 N a-Fe (BCC) 5.0 10 77 500 3.1 10-12 Y-Fe (FCCY 9.110 168 500 4.010 Self-Diffusion Fe a-Fe (BCC) 2.8 10 251 500 3.0 10-1 900 1.810-1 Fe Y-Fe (FCCY 5.0 10-5 284 900 1.110-17 1100 7.7 10- Cu Cu 2.5 107 200 500 7510 Al 2.310 144 500 Mg- Mg 1.5x10 136 500 Zn- Zn 1.5105 94 500 Mo 1.810 461 500 4.2 10-14 9.610 6.6 10-12 1.210 NI Ni 1.9x10 285 500 1.0 1033 Interdiffusion (Vacancy) Zn- Cu 2.4 10 189 500 4.0x108 Cu Zn 2.1x10 124 500 8.7 x 10 Cu Al 6.510 136 500 4.2 10 Mg- Al 1.2x10 Cu Ni 2.710 256 Ni Cu 1.9 10 1.9x10 1.310-22 5.4 10- Tabulation of Error Function Values 130 500 500 230 500 erf(z) erf(z) erf(z) 0 0.55 0.5633 0.9340 0.025 0.0282 0.60 0.6039 1.4 0.9523 0.05 0.0564 0.65 0.6420 1.5 0.9661 0.10 0.1125 0.70 0.6778 1.6 0.9763 0.15 0.1680 0.75 0.7112 1.7 0.9838 0.20 0.2227 0.80 0.7421 1.8 0.9891 0.25 0.2763 0.85 0.7707 1.9 0.9928 0.30 0.3286 0.90 0.7970 2.0 0.9953 0.35 0.3794 0.95 0.8209 2.2 0.9981 0.40 0.4284 1.0 0.8427 2.4 0.9993 0.45 0.4755 1.1 0.8802 2.6 0.9998 0.50 0.5205 12 0.9103 2.8 0.9999 Adario from Caller & Baby
Step by Step Solution
There are 3 Steps involved in it

Get step-by-step solutions from verified subject matter experts


