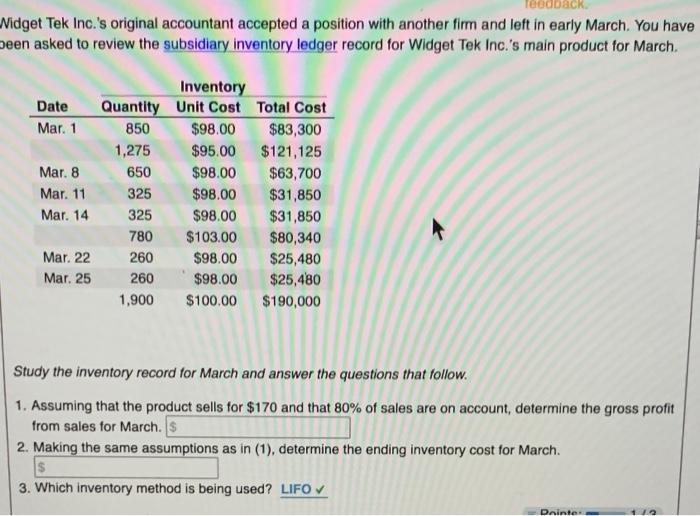
feedback. Widget Tek Inc.'s original accountant accepted a position with another firm and left in early...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
feedback. Widget Tek Inc.'s original accountant accepted a position with another firm and left in early March. You have been asked to review the subsidiary inventory ledger record for Widget Tek Inc.'s main product for March. Date Mar. 1 Mar. 8 Mar. 11 Mar. 14 Mar. 22 Mar. 25 Quantity 850 1,275 650 325 325 780 260 260 1,900 Inventory Unit Cost Total Cost $98.00 $95.00 $98.00 $98.00 $98.00 $103.00 $98.00 $98.00 $100.00 $83,300 $121,125 $63,700 $31,850 $31,850 $80,340 $25,480 $25,480 $190,000 Study the inventory record for March and answer the questions that follow. 1. Assuming that the product sells for $170 and that 80% of sales are on account, determine the gross profit from sales for March. $ 2. Making the same assumptions as in (1), determine the ending inventory cost for March. $ 3. Which inventory method is being used? LIFO ✔ Pointe feedback. Widget Tek Inc.'s original accountant accepted a position with another firm and left in early March. You have been asked to review the subsidiary inventory ledger record for Widget Tek Inc.'s main product for March. Date Mar. 1 Mar. 8 Mar. 11 Mar. 14 Mar. 22 Mar. 25 Quantity 850 1,275 650 325 325 780 260 260 1,900 Inventory Unit Cost Total Cost $98.00 $95.00 $98.00 $98.00 $98.00 $103.00 $98.00 $98.00 $100.00 $83,300 $121,125 $63,700 $31,850 $31,850 $80,340 $25,480 $25,480 $190,000 Study the inventory record for March and answer the questions that follow. 1. Assuming that the product sells for $170 and that 80% of sales are on account, determine the gross profit from sales for March. $ 2. Making the same assumptions as in (1), determine the ending inventory cost for March. $ 3. Which inventory method is being used? LIFO ✔ Pointe
Expert Answer:
Answer rating: 100% (QA)
A L 2 Part 1 3 Firstly we have to calculate the 4 cost of ... View the full answer

Related Book For 

Accounting Tools for Business Decision Making
ISBN: 978-1118128169
5th edition
Authors: Paul D. Kimmel, Jerry J. Weygandt, Donald E. Kieso
Posted Date:
Students also viewed these accounting questions
-
Sam is a LAW 960 student who drives a Nissan car. One of his classmates, Lulu, likes Sam's car very much and offered to purchase Sam's car for $20,000. Sam was reluctant to sell the car to Lulu, but...
-
Sam Milner, a lawyer, accepts a legal engagement in March, performs the work in April, and is paid in May. If Milners law firm prepares monthly financial statements, when should it recognize revenue...
-
Sam is a sole proprietor who owns, leases, and manages several apartment complexes and office buildings. During the current year, Sam incurs the following expenses. Which of these expenditures are...
-
You have been asked to calculate the cost of capital of Mulligan Ltd, a company which specialises in developing new medical equipment products. The following information is available regarding the...
-
Answer the following questions based on two assumptions: (1) Inflation increases the prices of all goods by 20%. (2) Ina's income increases from $50,000 to $55,000. a. Has Ina's budget line become...
-
Green Earth Landscaping Company provides monthly and weekly landscaping and maintenance services to residential customers in the tri-city area. Green Earth has no variable administrative expense....
-
For any pure substance, the difference between \(C_{P}\) and \(C_{V}\) can be expressed in terms of the isothermal compressibility \(\alpha\) and volume expansivity \(\beta\) as (a)...
-
Sharp Company has $15,000 to invest. The company is trying to decide between two alternative uses of the funds as follows: Sharp Company uses a 16% discount rate. Required: (Ignore income taxes.)...
-
Consider a 110 ft long, 1 in. type M copper tubing. (Due to the nature of this problem, do not use rounded intermediate values in your calculations-including answers submitt What is the outside...
-
At December 31, 2019, Catu Medical reported the following information on its statement of financial position. Accounts receivable R$960,000 Less: Allowance for doubtful accounts 80,000 During 2020,...
-
Despite Sherries initial reluctance, Josh ultimately prevailed on her to expand Fins regionally to Charlotte and Atlanta, but they agreed not to accept any outside equity investment. Instead, they...
-
Multiply and add like terms: x + x + 6 6 Question Help: Message instructor Calculator
-
Given A = -3 7 - 12 282 -5-20 find one nontrivial solution of Ax = 0 by inspection. [Hint: Think of the equation Ax = 0 written as X= (Type an integer or simplified fraction for each matrix element.)
-
Simplify the expression. 5-1/257/2
-
Graph the line. 25 y = -=x+2 -10 8 6 -co 6 9. 2 6 1D
-
1 -2 23-1 3) (15+10 p.) Given the matrix A = -3 6-11 7 2-4 58 4 (a) Find a basis for the column space of the matrix that consists of column vectors of A. (b) Find the rank and nullity of A.
-
Assume the money supply in a country triples.What happens to the nominal exchange rate if money is neutral? What are international reserves? Which currency is most commonly used in international...
-
Graph one period of each function. y = 4 cos x
-
The ledger of Wade Corporation at December 31, 2014, after the books have been closed, contains the following stockholders equity accounts. Preferred Stock (10,000 shares...
-
The notes that accompany a companys financial statements provide informative details that would clutter the amounts and descriptions presented in the statements. Refer to the financial statements of...
-
Pat Okendo started her own consulting firm, Okendo Consulting, on May 1, 2014. The trial balance at May 31 is as shown on the next page. In addition to those accounts listed on the trial balance, the...
-
Show that for a van der Waals gas, C V is a function only of temperature.
-
Justify the statement with mathematical expression: "For all the gases, the positive and negative values of the Joule-Thomson coefficient do not indicate the attainment of cooling effect and heating...
-
Prove that \(C_{P}-C_{V}=R\) for an ideal gas.

Study smarter with the SolutionInn App


