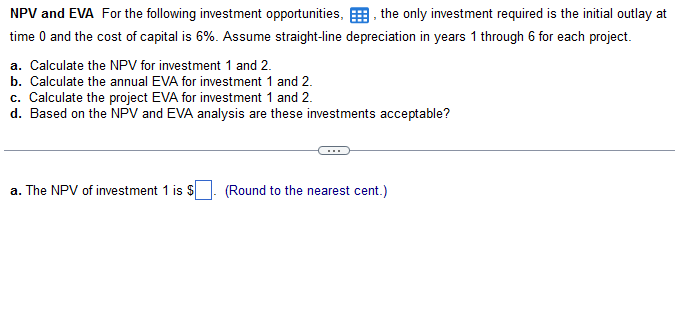
NPV and EVA For the following investment opportunities, the only investment required is the initial outlay...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
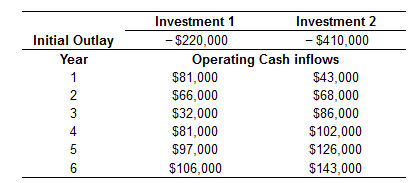
NPV and EVA For the following investment opportunities, the only investment required is the initial outlay at time 0 and the cost of capital is 6%. Assume straight-line depreciation in years 1 through 6 for each project. 2 a. Calculate the NPV for investment 1 and 2. b. Calculate the annual EVA for investment 1 and 2. c. Calculate the project EVA for investment 1 and 2. d. Based on the NPV and EVA analysis are these investments acceptable? a. The NPV of investment 1 is $ (Round to the nearest cent.) Initial Outlay Year 1 2345 6 Investment 1 - $220,000 Investment 2 - $410,000 Operating Cash inflows $81,000 $66,000 $32,000 $81,000 $97,000 $106,000 $43,000 $68,000 $86,000 $102,000 $126,000 $143,000 NPV and EVA For the following investment opportunities, the only investment required is the initial outlay at time 0 and the cost of capital is 6%. Assume straight-line depreciation in years 1 through 6 for each project. 2 a. Calculate the NPV for investment 1 and 2. b. Calculate the annual EVA for investment 1 and 2. c. Calculate the project EVA for investment 1 and 2. d. Based on the NPV and EVA analysis are these investments acceptable? a. The NPV of investment 1 is $ (Round to the nearest cent.) Initial Outlay Year 1 2345 6 Investment 1 - $220,000 Investment 2 - $410,000 Operating Cash inflows $81,000 $66,000 $32,000 $81,000 $97,000 $106,000 $43,000 $68,000 $86,000 $102,000 $126,000 $143,000
Expert Answer:
Answer rating: 100% (QA)
a Calculate the NPV for investment 1 and 2 The NPV is calculated by discounting each cash flow to its present value and summing them up The formula fo... View the full answer

Related Book For 

Finite Mathematics And Its Applications
ISBN: 9780137616619
13th Edition
Authors: Larry J. Goldstein, David I. Schneider, Martha J. Siegel , Jill Simmons
Posted Date:
Students also viewed these finance questions
-
Tasty Foods Corporation is a food conglomerate with major product lines of cereals, frozen dinners, and canned sodas and fruit juices. The firm was founded in 1965 by Henry Abercrombie, an ambitious...
-
The Think-Big Development Co. is a major investor in commercial real estate development projects. It currently has the opportunity to share in three large construction projects: Project 1: A...
-
The following additional information is available for the Dr. Ivan and Irene Incisor family from Chapters 1-5. Ivan's grandfather died and left a portfolio of municipal bonds. In 2012, they pay Ivan...
-
The Cano Company is decentralized, and divisions are considered investment centers. Cano has one division that manufactures oak dining room chairs with upholstered seat cushions. The Chair Division...
-
Which of the two asset-and-liability revenue recognition models currently being considered by the FASB and IASB is more similar to current practice?
-
The overall heat transfer coefficient of a wall is determined to be U = 0.075 Btu/h ft 2 F under the conditions of still air inside and winds of 7.5 mph outside. What will the U-factor be when the...
-
Petitioner Christy Brzonkala met respondents Antonio Morrison and James Crawford at a campus party at Virginia Polytechnic Institute (Virginia Tech), where they were all students. At the party, the...
-
A process currently services an average of 50 customers per day. Observations in recent weeks show that its utilization rate, utilization is about 90 percent, allowing for just a 10 percent capacity...
-
Calculate the following for a 3 ft 4 ft window with the label to the right. The summer design conditions for peak cooling load are 70 and 60% RH inside; 90 and 90% outside; and a peak direct solar...
-
First Trax Company manufactures snowboards. Its standard cost information follows. First Trax has the following actual results for the month of June: Number of units produced and sold...
-
Abel Weekend, Hanna Riri and Michelle Jackson are students living in Purple Rainier Residence Hall, at Centennial College. The residence was established in 1999 as a co-ed dormitory and is located...
-
A 1 5 - year mortgage for $ 3 7 0 , 0 0 0 with an interest rate of 5 . 1 % . ( HINT: You should first create and complete the TVM data table to compute the monthly payment. Then, this monthly payment...
-
How do communication theories such as social constructionism, symbolic interactionism, and discourse analysis inform our understanding of the complexities inherent in interpersonal, organizational,...
-
A division of the Gibson Corporation manufactures bicycle pumps. Each pump sells for $ 1 0 , and the variable cost of producing each unit is 6 0 % of the selling price. The monthly fixed costs...
-
Assume that the organisation to not subsribe to any standartds, framework and best practice, how this impact the IS audit conclusion and what impact it will have in the financial sector as financial...
-
The Rick Sanchez Corp recently reported $4,678,000 of sales, $3,740,500 of operating costs other than depreciation, and $450,2200 of depreciation. The company had $533,250 of outstanding bonds that...
-
A dia- Ils top- custo- rs who Bentley m for ations r. ollow- prop- entley d. a link table must be created to accept the for- eign keys of both tables. e. none of the above is true. PROBLEM 2: REA...
-
on 8 For the following set of lengths 130, 170, 160, 160, 150, 190 Third quartile is: et red d out of Select one: O a. 160 a question O b. 145 O c. 175 O d. 180
-
Define the intersection of the two sets A and B.
-
Find the x-intercept and the y-intercept of each line. y = -4x + 8
-
What are discount points?
-
Show that if a liquid is in equilibrium with its own vapour and an inert gas in a closed vessel, then \[\frac{\mathrm{d} p_{v}}{\mathrm{~d} p}=\frac{ho_{v}}{ho_{l}}\] where \(p_{v}\) is the partial...
-
a. Describe the meaning of the term thermodynamic equilibrium. Explain how entropy can be used as a measure of equilibrium and also how other properties can be developed which can be used to assess...
-
Show that when different phases are in equilibrium the specific Gibbs energy of each phase is equal. Using the following data, show the pressure at which graphite and diamond are in equilibrium at a...

Study smarter with the SolutionInn App


