Question: 1) Draw an electron-pushing mechanism for the formation of the propionylium cation resulting from the reaction of propionyl chloride and FeCl3. Show all lone
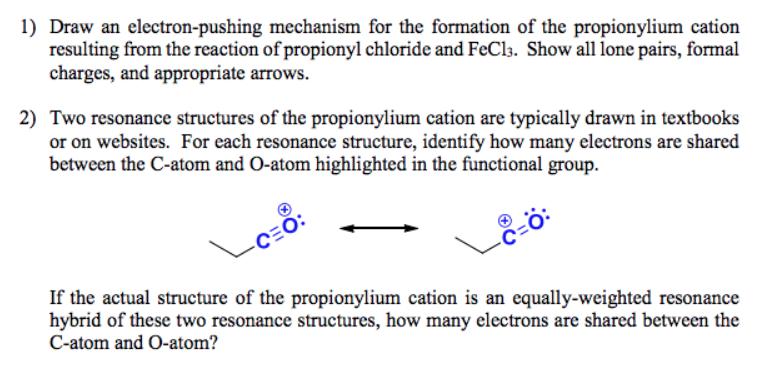
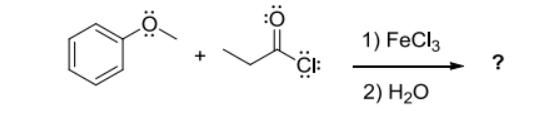
1) Draw an electron-pushing mechanism for the formation of the propionylium cation resulting from the reaction of propionyl chloride and FeCl3. Show all lone pairs, formal charges, and appropriate arrows. 2) Two resonance structures of the propionylium cation are typically drawn in textbooks or on websites. For each resonance structure, identify how many electrons are shared between the C-atom and O-atom highlighted in the functional group. CEC If the actual structure of the propionylium cation is an equally-weighted resonance hybrid of these two resonance structures, how many electrons are shared between the C-atom and O-atom? CE 1) FeCl3 2) HO ?
Step by Step Solution
3.51 Rating (161 Votes )
There are 3 Steps involved in it
ANSWER CI FeCl3 6 e... View full answer

Get step-by-step solutions from verified subject matter experts


