Of the N2O4 molecules present in a sample of the gas, 12.5% are dissociated into NO2...
Fantastic news! We've Found the answer you've been seeking!
Question:
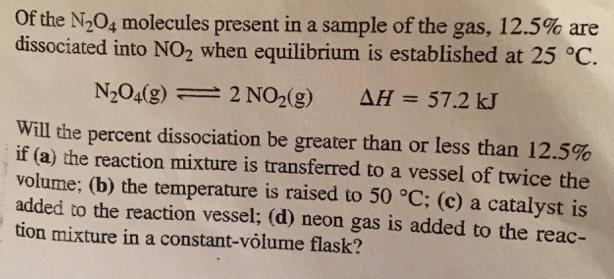
Transcribed Image Text:
Of the N2O4 molecules present in a sample of the gas, 12.5% are dissociated into NO2 when equilibrium is established at 25 °C. N2O4(g) = 2 NO2(g) AH = 57.2 kJ Will the percent dissociation be greater than or less than 12.5% if (a) the reaction mixture is transferred to a vessel of twice the volume; (b) the temperature is raised to 50 °C; (c) a catalyst is added to the reaction vessel; (d) neon gas is added to the reac- tion mixture in a constant-volume flask? Of the N2O4 molecules present in a sample of the gas, 12.5% are dissociated into NO2 when equilibrium is established at 25 °C. N2O4(g) = 2 NO2(g) AH = 57.2 kJ Will the percent dissociation be greater than or less than 12.5% if (a) the reaction mixture is transferred to a vessel of twice the volume; (b) the temperature is raised to 50 °C; (c) a catalyst is added to the reaction vessel; (d) neon gas is added to the reac- tion mixture in a constant-volume flask?
Expert Answer:
Answer rating: 100% (QA)
A greater than 125 since volume has been increased so pressure will decrease to maintain the equilib... View the full answer

Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
Is the mode field radius greater than or less than the fiber core radius in single-mode step index fiber?
-
In a sample of 900 gas stations, the mean price of regular gasoline at the pump was $3.746 per gallon and the standard deviation was $0.009 per gallon. A random sample of size 55 is selected from...
-
max for the * transition in ethylene is 170 nm. Is the HOMOLUMO energy difference in ethylene greater than or less than that of cis, trans-1,3-cyclooctadiene?
-
Population increase in northern Virginia requires new road constructions and repairs annually from 2022. More public schools will also be needed by 2035. The state of Virginia is considering an...
-
Let n,k Z+, and define p(n, k) to be the number of partitions of n into exactly k (positive-integer) summands. Prove that p(n, k) = p(n - 1), (k - 1) + p(n - k, k).
-
You can determine wind direction by wetting your finger and holding it up in the air. Explain.
-
Mr. Tam is the front man for the Asian American dance-rock band The Slants. Mr. Tam named his band The Slants to reclaim and take ownership of Asian stereotypes. The band draws inspiration for its...
-
Loveless Corporation had the following 2010 income statement. Revenues............................$100,000 Expenses.................................60,000...
-
Arbon Company has three service departments and two operating departments. Selected data concerning the five departments are presented below: Costs Number of employees Square metres of space occupied...
-
Top Quality Appliance-Long Beach has just purchased a franchise from Top Quality Appliance (TQA). TQA is a manufacturer of kitchen appliances. TQA markets its products via retail stores that are...
-
A common argument against the transitivity property of preferences is that people cannot discern between small differences, but, then assuming transitivity implies that they should be indifferent...
-
In Ron Howard's Ted Talk, How I discovered the black art of cult branding, he lists Six major cult brand tactics that matter most when building brands and targeting customers. How I discovered the...
-
What are the key principles underlying the O(1) scheduler in the Linux kernel, and how does it achieve constant-time scheduling decisions regardless of the number of processes in the system?
-
Suppose one Euro can purchase 1.25 U.S. dollars today in the foreign exchange market, and currency forecasters predict that the U.S. dollar will appreciate by 25% against the Euro over the next 60...
-
At what point should we stop a Fishbone Diagram during its analysis? What problems can be foreseen in an NGT way of attaining a decision? Can Delphi technique be used to help with NGT? Can Project...
-
You learned that both the state and federal governments have policies in place to encourage marriage. Despite this, the states have put various requirements and restrictions on getting married. Some...
-
What strategies can be employed to foster a culture of ethical mindfulness and moral courage within organizations, enabling employees to navigate ethical challenges with integrity and moral...
-
Find the intercepts and then graph the line. (a) 2x - 3y = 6 (b) 10 - 5x = 2y
-
Dinitrogen tetroxide, N2O4, is a colorless gas (boiling point, 21oC), which dissociates to give nitrogen dioxide, NO2, a reddish brown gas. The equilibrium constant Kc at 25oC is 0.125. What...
-
Some lemon juice has a hydronium-ion concentration of 5.0 10-3 M. What is the pH of the lemon juice?
-
Which of the following has the largest number of molecules? a. 1 g of benzene, C 6 H 6 b. 1 g of formaldehyde, CH 2 O c. 1 g of TNT, C 7 H 5 N 3 O 6 d. 1 g of naphthalene, C 10 H 8 e. 1 g of glucose,...
-
What makes channel stuffing illegal by comparison to a car dealership that runs mega end-of-year sales?
-
How would an audit not catch missing cash amounts of this magnitude?
-
While Al Capone was clearly the leader of his organization and benefited extensively, what enticed others to participate in his illegal activities?

Study smarter with the SolutionInn App


