what is the ED(destabilized energy) for chairs and its percentage minor and major isomer in equilibrium...
Fantastic news! We've Found the answer you've been seeking!
Question:
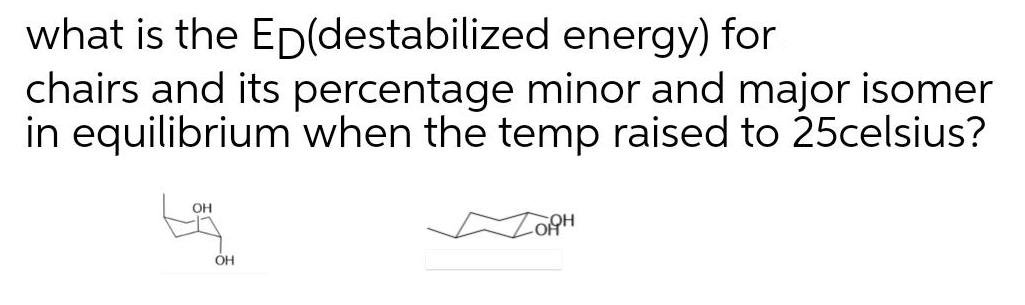
Transcribed Image Text:
what is the ED(destabilized energy) for chairs and its percentage minor and major isomer in equilibrium when the temp raised to 25celsius? OH what is the ED(destabilized energy) for chairs and its percentage minor and major isomer in equilibrium when the temp raised to 25celsius? OH
Expert Answer:
Answer rating: 100% (QA)
EDdestablization energy for extremely left side chair conformation inwhich two OH groups are in axia... View the full answer

Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
The equilibrium between hydrogen cyanide (HCN) and its isomer hydrogen isocyanide (HNC) is important in interstellar chemistry: HCN (g) HNC (g) A long-standing puzzle regarding this reaction is that...
-
What red flag is raised when a company has an unbilled contract revenue account in its general ledger?
-
Ed is undergoing surgery for an old football injury and must be anesthetized. The anesthesiologist knows Ed will be "under" when the concentration of sodium pentathol in his blood is at least 50...
-
In Figure P23.15, determine the point (other than infinity) at which the electric field is zero. 1.00 m -2.50 : 6.00 C
-
A principle of microeconomics instructor regularly asks her class to give an example of an inferior good. No matter how poor we might be, the students tell her, ramen noodles are an inferior good....
-
Utica Manufacturing (UM) was recently acquired by MegaMachines, Inc. (MM), and organized as a separate division within the company. Most manufacturing plants at MM use an ABC system, but UM has...
-
Steven Sanchez worked as a warehouse employee for Gruma Corporation. In December 2016, Sanchez was presented an arbitration agreement during a meeting with George Tate, the human resources manager...
-
Bug-Off Exterminators provides pest control services and sells extermination products manufactured by other companies. The following six-column table contains the companys unadjusted trial balance as...
-
Why would you perform a silent installation? What is the purpose of a password complexity policy? Oracle offers a free download of all editions of the Oracle Database. How does the company derive...
-
The eight members of the Model United Nations team from Pima Community College are lining up for a photo. a) How many different ways can the eight team members line up? b) What is the probability...
-
Compare and comment on the number of exceptions for both the S&P500 Index and the Shanghai Composite Index. From your comparison, is it reasonable to use the VaR model for the Chinese market compared...
-
In this circuit, the battery has a voltage of 12 V between terminals (and a negligible internal resistance). We are told that the resistor R in the middle branch of the circuit would dissipate 1.5 W...
-
On March 1, XYZ Company bought $150 worth of supplies. At the end of March, the company had used $100 of these supplies in the business. a) What amount of expense should the company record on the...
-
At the start of the year, deft records purchases a new piece of recording equipment for $7,000. deft expects to use the equipment for 5 years and then to sell it for $500 . at the end of two years,...
-
Which of the following statements is correct? a. The stronger the linear relationship between two variables, the closer the correlation coefficient will be to 1.0. b. Two variables that are...
-
"How does the interplay between social structure and social interactions evolve over time, and what implications does this dynamic relationship have for society?"
-
A ski-jumper slides down a sloping ramp. At the bottom part of the ramp is an upward curve to the takeoff point at the end of the ramp. The start point is a vertical height of 25 m above the takeoff...
-
What are technical skills At what level are they most important and why?
-
Given the following kinetic scheme and associated rate constants, determine the concentration profiles of all species using Eulers method. Assume that the reaction is initiated with only the reactant...
-
Electronic spectroscopy of the HgAr van der Waals complex was performed to determine the dissociation energy of the complex in the first excited state (Quayle, C. J. K. et al. Journal of Chemical...
-
Why can more work be extracted from a fuel cell than a combustion engine for the same overall reaction?
-
Over what ranges of taxable income in 2017 will the total income tax liability for two persons with equal incomes who file as single individuals equal their income tax liability if they file jointly...
-
If a taxpayer has $40,000 of employee salary in 2017, how much will be withheld for the Social Security and Medicare taxes?
-
Sally and Jim are married and have taxable income in 2017 of $160,000. If they could file their income tax as single individuals, each of them would have taxable income of $80,000. Do they have a...

Study smarter with the SolutionInn App


