sample weight (gms) 2. What osmotic pressure (77) would be developed across the semipermeable membrane of...
Fantastic news! We've Found the answer you've been seeking!
Question:

Transcribed Image Text:
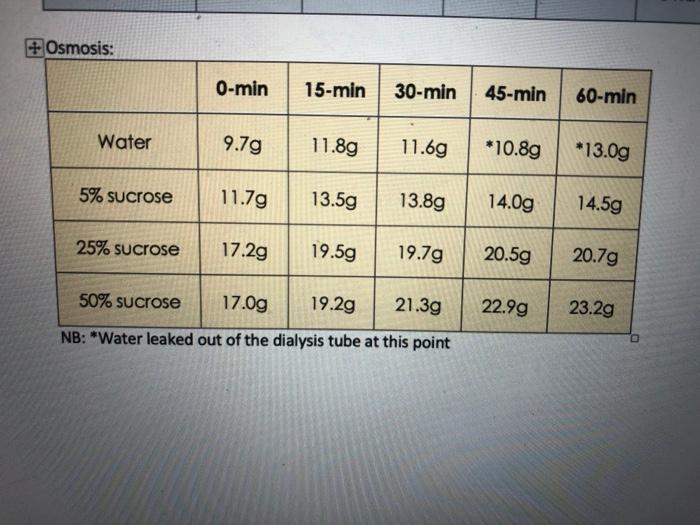
sample weight (gms) 2. What osmotic pressure (77) would be developed across the semipermeable membrane of a sample containing: a. 5% sucrose b. 0.9 % NaCl c. 2 M CaCl₂ 3. a. Is the rate of osmosis constant? Why? GOU 50'.. 1. Results tube # 1. b. What is the relationship between the concentration of sucrose in each bag and the rate of osmosis? The relatenship between the concentration of sucrese in each vay and the rate of osmosis is . Tonicity hct 2 solution tonicity response (lysis, crenation, etc.) +Osmosis: Water 11.6g *13.0g 13.8g 14.0g 14.5g 19.5g 19.7g 20.5g 20.7g 17.0g 19.2g 21.3g NB: "Water leaked out of the dialysis tube at this point 5% sucrose 25% sucrose 0-min 15-min 30-min 45-min 60-min 50% sucrose 9.7g 11.8g 11.7g 13.5g 17.2g *10.8g 22.99 23.2g sample weight (gms) 2. What osmotic pressure (77) would be developed across the semipermeable membrane of a sample containing: a. 5% sucrose b. 0.9 % NaCl c. 2 M CaCl₂ 3. a. Is the rate of osmosis constant? Why? GOU 50'.. 1. Results tube # 1. b. What is the relationship between the concentration of sucrose in each bag and the rate of osmosis? The relatenship between the concentration of sucrese in each vay and the rate of osmosis is . Tonicity hct 2 solution tonicity response (lysis, crenation, etc.) +Osmosis: Water 11.6g *13.0g 13.8g 14.0g 14.5g 19.5g 19.7g 20.5g 20.7g 17.0g 19.2g 21.3g NB: "Water leaked out of the dialysis tube at this point 5% sucrose 25% sucrose 0-min 15-min 30-min 45-min 60-min 50% sucrose 9.7g 11.8g 11.7g 13.5g 17.2g *10.8g 22.99 23.2g
Expert Answer:
Answer rating: 100% (QA)
iCRT iMRT inrtv where M molarity n moles v vol in litre ... View the full answer

Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
A salt solution has an osmotic pressure of 17 atmospheres at 22C. What is the freezing point of this solution? What assumptions must be made to solve this problem?
-
Water leaked from a tank at a rate of r(t) liters per hour, where the graph of is as shown. Use Simpsons Rule to estimate the total amount of water that leaked out during the first six hours. 2 4
-
What is the osmotic pressure of a 0.0522 M solution of C12H22O11 at 55C?
-
9. What is the coordination entity formed when excess of aqueous KCN is added to an aqueous solution of copper sulphate? Why is it that no precipitate of copper sulphide is obtained when H2S(g) is...
-
The BOM for product A is shown in Figure. The MPS for product A calls for 120 units to be started in weeks 2, 4, 5, and 8. Table shows data from the inventory records. Develop the material...
-
What is the difference between a team and a group? Describe your personal experience with each.
-
Spice Inc. issued bonds payable on December 31. Spice's bonds were dated July 31. Which statement is true of Spice's journal entry to record issuance of the bonds payable? a. Spice must pay one...
-
On March 31, 2018, Chow Brothers, Inc., bought 10% of KT Manufacturing's capital stock for $50 million. KT's net income for the year ended December 31, 2018, was $80 million. The fair value of the...
-
The transformation steps from your EER Model (Appendix A) to your final tables (Task 1); Make sure to show each step of the transformation, and the final transformation tables; The DDL...
-
Pequity Company purchased 85% of the common stock of Sequity Company on April 1, Year 1 for total consideration of $545,000 cash plus $50,000 of contingent consideration as measured according to GAAP...
-
Consider two identical metal spheres, A and B. Sphere A carries a charge of - 48 C and sphere B carries a charge of - 64 C. The two spheres are touched together and then separated after they acquire...
-
What is the current account? Use the information in the following table on Mexico's 2007 international transactions to answer exercises 4-6 (the amounts are the U.S. dollar values in millions):...
-
A test is made of H0 : = 100 versus H1: 100. The sample mean is x = 97, the sample size is n = 75, and the population standard deviation is = 8. a. Find the value of the test statistic z. b. Find...
-
For each of the following P-values, state whether the result is statistically significant at the 0.10 level. a. P = 0.08 b. P = 0.15 c. P = 0.01 d. P = 0.50
-
A hypothesis test is performed with a significance level of = 0.05. a. If the P-value is 0.08, is H0 rejected? b. If the P-value is 0.08, are the results statistically significant at the 0.05 level?...
-
A test is made of H0 : = 30 versus H1: < 30. The test statistic is z = 1.28. Find and interpret the P-value.
-
D D Question 19 Which person, when elected to the U.S. Senate in 1961, rejuvenated the Republican Party in Texas? Lloyd Bentsen Phil Gramm John Tower O Kay Bailey Hutchinson Question 20 What is the...
-
A supermarket chain is interested in exploring the relationship between the sales of its store-brand canned vegetables (y), the amount spent on promotion of the vegetables in local newspapers (x1)...
-
Volatile organic compounds (VOCs) can be removed from water effluents by stripping in packed towers. Possible stripping agents are steam and air. Alternatively, the VOCs can be removed by carbon...
-
Exit gas from a chlorinator consists of a mixture of 20 mol% chlorine in air. This concentration is to be reduced to 1% chlorine by water absorption in a packed column to operate isothermally at 20C...
-
Benzoic acid is to be crystallized by bulk-phase desublimation from N2 using a novel method described by Vitovec, Smolik, and Kugler [Coll. Czech. Chem. Commun., 42, 1108-1117 (1977)]. The gas,...
-
A reversible process is a process (a) Which proceeds with no driving force (b) Which takes place spontaneously (c) Which is quasi-static (d) Which is frictional process.
-
At constant temperature and pressure, the free energy for a chemically reacting system at equilibrium is (a) Minimum (b) Maximum (c) Can not be predicted (d) None of these.
-
The operation of a throttling device follows the (a) Zeroth law of thermodynamics (b) First law of thermodynamics (c) Second law of thermodynamics (d) Third law of thermodynamics.

Study smarter with the SolutionInn App


