Match each of the following ions and molecules with its correct molecular geomet IC1 NH CIO3...
Fantastic news! We've Found the answer you've been seeking!
Question:


Transcribed Image Text:
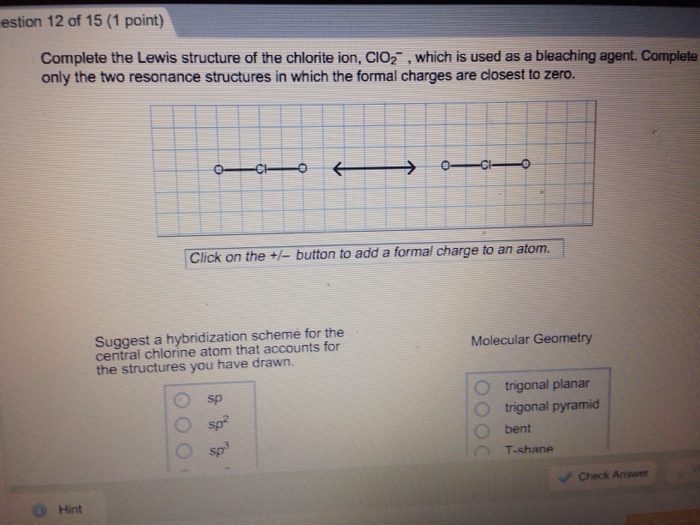
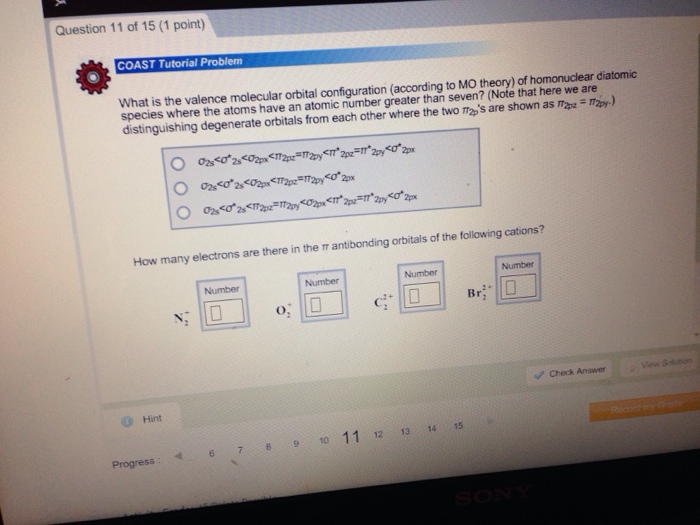
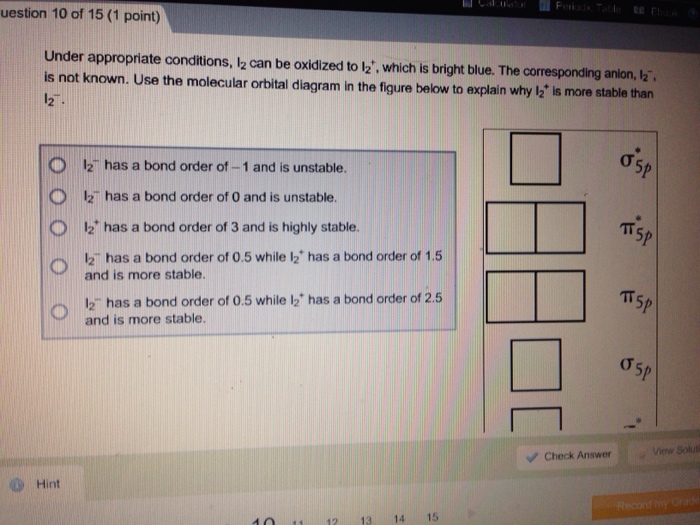
Match each of the following ions and molecules with its correct molecular geomet IC1₂ NH₂ CIO3 BC tetrahedral trigonal pyramidal linear trigonal planar estion 12 of 15 (1 point) Complete the Lewis structure of the chlorite ion, CIO₂, which is used as a bleaching agent. Complete only the two resonance structures in which the formal charges are closest to zero. Hint 1 C Click on the +/- button to add a formal charge to an atom. Suggest a hybridization scheme for the central chlorine atom that accounts for the structures you have drawn. O CI sp sp² sp³ Molecular Geometry trigonal planar trigonal pyramid bent T-shane Check Answer Question 11 of 15 (1 point) COAST Tutorial Problem What is the valence molecular orbital configuration (according to MO theory) of homonuclear diatomic species where the atoms have an atomic number greater than seven? (Note that here we are distinguishing degenerate orbitals from each other where the two 's are shown as ap=2py) Hint 02550*2<02px<112p=112y<112p=112p<0 2px 028<0*28 <02px<112p=112py 02 <0*26 <112p=112p<0x<12p=11* 2y<0*2px How many electrons are there in the antibonding orbitals of the following cations? Progress: Number 10 7 8 <o²2px o; 0 9 Number Number 10 10 11 12 13 14 15 Number Br 0 Check Answer uestion 10 of 15 (1 point) Under appropriate conditions, 12 can be oxidized to 12₂, which is bright blue. The corresponding anion, 1₂. is not known. Use the molecular orbital diagram in the figure below to explain why 12 is more stable than 12. Hint 12 has a bond order of -1 and is unstable. 12 has a bond order of 0 and is unstable. 12 has a bond order of 3 and is highly stable. 12 has a bond order of 0.5 while 12 has a bond order of 1.5 and is more stable. 12 has a bond order of 0.5 while l2 has a bond order of 2.5 and is more stable. 11 12 13 14 Periodic Table Ebook 15 Check Answer OSP TTSP SP 05P View Soluti Calculator Periodic Table RE on 9 of 15 (1 point) Cyclic structures exist for many compounds of carbon and hydrogen. Describe the hybridization aroun each carbon atom in benzene (C6H6), cyclobutane (C4H8), and cyclobutene (C4H6). Match each of the following ions and molecules with its correct molecular geomet IC1₂ NH₂ CIO3 BC tetrahedral trigonal pyramidal linear trigonal planar estion 12 of 15 (1 point) Complete the Lewis structure of the chlorite ion, CIO₂, which is used as a bleaching agent. Complete only the two resonance structures in which the formal charges are closest to zero. Hint 1 C Click on the +/- button to add a formal charge to an atom. Suggest a hybridization scheme for the central chlorine atom that accounts for the structures you have drawn. O CI sp sp² sp³ Molecular Geometry trigonal planar trigonal pyramid bent T-shane Check Answer Question 11 of 15 (1 point) COAST Tutorial Problem What is the valence molecular orbital configuration (according to MO theory) of homonuclear diatomic species where the atoms have an atomic number greater than seven? (Note that here we are distinguishing degenerate orbitals from each other where the two 's are shown as ap=2py) Hint 02550*2<02px<112p=112y<112p=112p<0 2px 028<0*28 <02px<112p=112py 02 <0*26 <112p=112p<0x<12p=11* 2y<0*2px How many electrons are there in the antibonding orbitals of the following cations? Progress: Number 10 7 8 <o²2px o; 0 9 Number Number 10 10 11 12 13 14 15 Number Br 0 Check Answer uestion 10 of 15 (1 point) Under appropriate conditions, 12 can be oxidized to 12₂, which is bright blue. The corresponding anion, 1₂. is not known. Use the molecular orbital diagram in the figure below to explain why 12 is more stable than 12. Hint 12 has a bond order of -1 and is unstable. 12 has a bond order of 0 and is unstable. 12 has a bond order of 3 and is highly stable. 12 has a bond order of 0.5 while 12 has a bond order of 1.5 and is more stable. 12 has a bond order of 0.5 while l2 has a bond order of 2.5 and is more stable. 11 12 13 14 Periodic Table Ebook 15 Check Answer OSP TTSP SP 05P View Soluti Calculator Periodic Table RE on 9 of 15 (1 point) Cyclic structures exist for many compounds of carbon and hydrogen. Describe the hybridization aroun each carbon atom in benzene (C6H6), cyclobutane (C4H8), and cyclobutene (C4H6).
Expert Answer:
Answer rating: 100% (QA)
In IC15 the molecule has 7271 22 valence electrons The three l... View the full answer

Related Book For 

Introductory Statistics Exploring The World Through Data
ISBN: 9780321978271
2nd Edition
Authors: Robert Gould, Colleen Ryan
Posted Date:
Students also viewed these chemistry questions
-
3 4 5 6 7 8 9 10 11 12 13 A 14 Cost of the Asset 15 Life of the Asset in Years 16 Book Value of the Asset after 5 years 17 Depreciable Basis 18 Yearly depreciation 19 After tax Salvage Value in year...
-
A wooden board AB, which is used as a temporary prop to support a small roof, exerts at point A of the roof a 228-N force directed along BA. Determine the moment about C of that force. 0.12 m D. 18m
-
In the secretory pathway, match each of the following terms with its description below. ____ free ribosome ____ signal peptide ____ signal recognition particle (SRP) ____ core glycosylation ____...
-
Tell whether the given side lengths form a right triangle. 8, 10, and 14
-
Suburban Heating, Inc., installs heating systems in new homes built in suburban Philadelphia. Jobs are priced using the time and materials method. The following predictions pertain to the companys...
-
An experiment has three steps with three outcomes possible for the first step, two outcomes possible for the second step, and four outcomes possible for the third step. How many experimental outcomes...
-
In the spectrometer shown in Figure P24.67, do the ions have positive or negative charge? A. Positive B. Negative. If you have a sample of unknown composition, a first step at analysis might be a...
-
Accountants at the Tucson firm, Larry Youdelman, CPAs, believed that several traveling executives were submitting unusually high travel vouchers when they returned from business trips. First, they...
-
Beech Corporation is a merchandising company that is preparing a master budget for the third quarter. The company's balance sheet as of June 30th is shown below: Assets Cash Beech Corporation Balance...
-
Presented below is information related to the sole proprietorship of Helen Archer, attorney. Legal service revenue2020 ...........$330,000 Total expenses2020 ........................211,000 Assets,...
-
Assume put premium=$5, S = $50, K=55, r = 5% p.a. continuously compounded, the stock pays a 3.0% p.a. continuous dividend and the time to maturity is 1 year. What is the implied volatility?
-
A sales associate is a licensed sales associate of a broker. By issuance of a Final Order, the broker's license was revoked by the Florida Real Estate Commission. As to the sales associate's license,...
-
The polynomial function q(x) = 2x 9x 12x +29x + 30 has known factors (x-2) and (x+1). Which expression represents q(x) as the product of linear factors? O (2x-5)(x+3)(x-2)(x+1) O...
-
What are the advantages of native app compared to the web app? Mobile Web app: Application built to run on the mobile web browser built into a smartphone or tablet computer - Tools: Web technologies...
-
The _ _ init _ _ method is used to initialize what components of an object? a . ) Its methods b . ) Its attributes c . ) Its scope d . ) Its structure
-
Suppose that f(x) is a continuous even function with What is the value of the integral below? Provide your answer below: XS 1(20) dir 9. f(x) da
-
Uneven cash flows: An investment costs $465 and is expected to produce cash flows of $100 at the end of each of the next 4 years, then an extra lump sum payment of $200 at the end of the fourth year....
-
Suppose the market is semistrong form efficient. Can you expect to earn excess returns if you make trades based on? a. Your brokers information about record earnings for a stock? b. Rumors about a...
-
Professors of ethics (Eth), professors of philosophy (Phil), and professors in fields other than philosophy or ethics (Other) were asked what percentage of income they actually donated to charity....
-
Suppose a church is considering purchasing new hymnals (books that contain the hymns sung during the service) and wants to sample the congregation to see whether its members sup port the purchase....
-
When the draft lottery for military service in the Vietnam War was conducted, officials "randomly" selected birthdays. For example, September 14 was selected first, and that date was assigned the...
-
The following information is available for McKee Corporation for 2015. 1. Excess of tax depreciation over book depreciation, 40,000. This 40,000 difference will reverse equally over the years...
-
In 2015, Amirante Corporation had pretax financial income of $168,000 and taxable income of $120,000. The difference is due to the use of different depreciation methods for tax and accounting...
-
Brennan Corporation began 2015 with a $90,000 balance in the Deferred Tax Liability account. At the end of 2015, the related cumulative temporary difference amounts to $350,000, and it will reverse...

Study smarter with the SolutionInn App


