Suppose you have a model: Y = X + where the observations are iid but & is
Fantastic news! We've Found the answer you've been seeking!
Question:
Suppose you have a model: Y = Xβ + ε
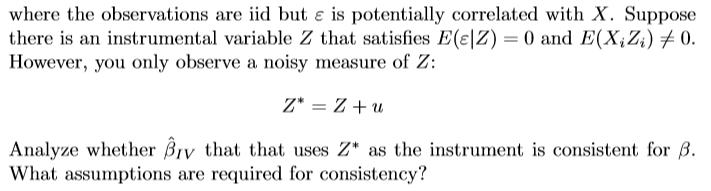
Transcribed Image Text:
€ where the observations are iid but & is potentially correlated with X. Suppose there is an instrumental variable Z that satisfies E(e|Z) = 0 and E(X₁Z₁) 0. However, you only observe a noisy measure of Z: Z* = Z+u Analyze whether By that that uses Z* as the instrument is consistent for 3. What assumptions are required for consistency? € where the observations are iid but & is potentially correlated with X. Suppose there is an instrumental variable Z that satisfies E(e|Z) = 0 and E(X₁Z₁) 0. However, you only observe a noisy measure of Z: Z* = Z+u Analyze whether By that that uses Z* as the instrument is consistent for 3. What assumptions are required for consistency?
Expert Answer:
Answer rating: 100% (QA)
Analy ze whether iv that that uses Z as the instrument is consistent for B ANS WER There is a pote... View the full answer

Related Book For 

Financial Markets And Institutions
ISBN: 978-0132136839
7th Edition
Authors: Frederic S. Mishkin, Stanley G. Eakins
Posted Date:
Students also viewed these economics questions
-
Below is information related to UMPI Pro Shops purchases of golf balls during April During the same month 1050 golf balls were sold UMPI Pro Shop uses the periodic inventory system Units Unit Cost...
-
The company has bonds outstanding with a face value of $50,000 and an unamortized premium of $2,350 at the beginning of the year and $2,000 as of the end of the year. Sales (all for cash) were...
-
A bond with a face value of $ 280,000 and a quoted price of 92 has a selling price of?
-
Assuming that the Fed judges inflation to be the most significant problem in the economy and that it wishes to employ all of its policy instruments except interest on reserves, what should the Fed do...
-
For the data of Problem 26.19, calculate slope intercepts and determine the end area by the coordinate method.
-
If d1 = 2a, determine the distance a and the centroidal polar moment of inertia of the 24- in2 shaded area shown knowing that d2 = 2in. and that the polar moments of inertia of the area with respect...
-
On February 20, 2009, Cedar Valley Aviation, a wholly owned subsidiary of Aerial Services, Inc. (ASI), brought a Piper 522AS (Cheyenne II) in for maintenance to Des Moines Flying Service, Inc....
-
Wild Wheels, Inc., wholesales a line of custom mountain bikes. Wild Wheels inventory, as of November 30, 201 2, consisted of 20 mountain bikes costing $550 each. Wild Wheels trial balance as of...
-
Antelopes, native to Africa and Asia, range in size from 30 cm to over 180 cm at the shoulder, with most between 90-120 cm. This is related to the humerus length. The data below gives the length and...
-
Tracy Morgan is self-employed as a professional golf instructor. She uses the cash method of accounting and her SS# is 465-34-3389. Her principal business code is 812990. Tracy's business is located...
-
Maryam has been hired as an intern at Samry, a small custom embroidery and design business. She is amazed at the many uses her manager has found for technology. She is able to draft customer designs...
-
2 NO5(g) 4NO (g) + 0(g) Concentration (M) Time(s) NO5 NO2 0 0 0.0200 0 0 100 0.0169 0.0063 0.0016 200 0.0142 0.0115 0.0029 0.0120 0.0160 0.0040 0.0101 0.0197 0.0049 0.0086 0.0229 0.0057 300 400 500...
-
Provide a proper general functional group formula and an example structure of each of the functional groups listedbelow with the IUPAC name. It would be useful for you to order the functional groups...
-
Would you expect nonpolar or polar molecules of similar mass to be more easily detected? Why?
-
Convert 0 . 0 6 2 mol Se to g Se . 2 . How many moles of CaF 2 are equal to 2 2 . 8 4 g CaF 2 ? 3 . Convert 5 . 2 x 1 0 2 4 atom Co to mol Co . 4 . How many molecules of PCl 3 are equal to 0 . 0 0 6...
-
write the one letter code for the oligopeptide thr - his - ile - ser - ile - ser - glu - ala - ser - tyr and calculate the total charge on it at pH 9 . 7 side chain pkA values 4 . 0 , Glu 5 . 0 , His...
-
During take-off a 5kg model rocket is burning fuel causing its speed to increase at a rate of 5m/s despite experiencing a 80N drag. Determine all unknowns. Are all forces on the rocket balanced?...
-
Kenneth Hubbard has prepared the following list of statements about managerial accounting and financial accounting. 1. Financial accounting focuses on providing information to internal users. 2....
-
A bank currently holds $150,000 in excess reserves. If the current reserve requirement is 12.5%, how much could the money supply change? How could this happen?
-
What does the month-end balance sheet for NewBank look like? Calculate this before any income tax consideration.
-
A swap agreement calls for Durbin Industries to pay interest annually based on a rate of 1.5% over the one-year T-bill rate, currently 6%. In return, Durbin receives interest at a rate of 6% on a...
-
Use Table 15.1 to find the short rate in the fourth year. Confirm that the discount factor on the 4-year zero is a geometric average of 1 + the short rates in the next four years. Table 15.1...
-
Using the data in Table 15.1, calculate the price and yield to maturity of a 3-year bond with a coupon rate of 4% making annual coupon payments. Does its yield match that of either the 3-year zero or...
-
Suppose that the required liquidity premium for the short-term investor is 1%. What must E(r2) be if f2 is 7%?

Study smarter with the SolutionInn App


