Metal cations in solution are complexed to HO molecules and exist as hydrated ions. For example,...
Fantastic news! We've Found the answer you've been seeking!
Question:
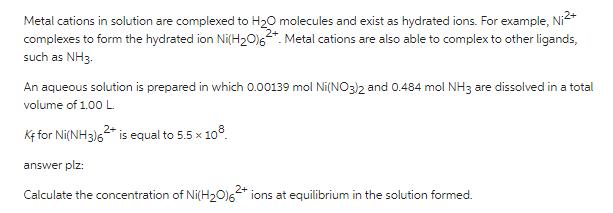
Transcribed Image Text:
Metal cations in solution are complexed to H₂O molecules and exist as hydrated ions. For example, Ni²+ 2+ complexes to form the hydrated ion Ni(H₂0)62. Metal cations are also able to complex to other ligands, such as NH3. An aqueous solution is prepared in which 0.00139 mol Ni(NO3)2 and 0.484 mol NH3 are dissolved in a total volume of 1.00 L. Kf for Ni(NH3)62* is equal to 5.5 x 108 answer plz: Calculate the concentration of Ni(H₂O)62+ ions at equilibrium in the solution formed. Metal cations in solution are complexed to H₂O molecules and exist as hydrated ions. For example, Ni²+ 2+ complexes to form the hydrated ion Ni(H₂0)62. Metal cations are also able to complex to other ligands, such as NH3. An aqueous solution is prepared in which 0.00139 mol Ni(NO3)2 and 0.484 mol NH3 are dissolved in a total volume of 1.00 L. Kf for Ni(NH3)62* is equal to 5.5 x 108 answer plz: Calculate the concentration of Ni(H₂O)62+ ions at equilibrium in the solution formed.
Expert Answer:
Answer rating: 100% (QA)
To calculate the concentration of NiH2O2 ions at equilibrium in the solution formed we need to use ... View the full answer

Related Book For 

Posted Date:
Students also viewed these accounting questions
-
Calculate the pH of a solution prepared by mixing 0.0800 mol of chloroacetic acid plus 0.0400 mol of sodium chloroacetate in 1.00 L of water. (a) First do the calculation by assuming that the...
-
n3 ||| -d/2 n>n>n neff || d/2 1 n In a general three-layer slab waveguide with central thickness d, the index step profile is shown as left, and n>n>n. The solution is in the form: Um = Aexp[r(x +...
-
Identify two of GE's recent acquisitions and summarizing the key details of each. Next, address the following two topics with respect to each acquisition: Which of the reasons (if any) for...
-
To save for her newborn son's college education, Lea Wilson will invest $1,000 at the beginning of each year for the next 18 years. The interest rate is 12 percent. What is the future value? 1)...
-
Riuto Company accumulated the following standard cost data concerning product I-Tal. Materials per unit: 2 pounds at $5 per pound Labor per unit: 0.2 hours at $14 per hour Manufacturing overhead:...
-
Examine the loan amortization table for the last 5 months of a $500,000, 15-year mortgage with an APR of 4.05%. Determine the missing table amounts. Payment Beginning Number Balance 176 $18,368.50...
-
What levels of an organization benefit most from a data warehouse?
-
The contribution format income statement for Westex, Inc., for its most recent period is given below: The company had average operating assets of $500,000 during the period. Required: 1. Compute the...
-
A manufacturer uses activity-based costing to assign overhead cost to products. Budgeted cost information for its activities follows Activity Purchasing Factory services Setup Budgeted Cost $ 175,000...
-
Pulsar Optics produces medical lasers for use in hospitals. The accounts and their balances appear in the ledger of Pulsar Optics on April 30 of the current year as follows: Preferred 1% Stock, $120...
-
The owner of GROC Inc. has had a busy three years. The owner is curious about her company's performance as she compares its financial statements. She is interested in bringing on an investor and...
-
1) A ball is thrown vertically upward with a speed of 15 miles per hour. How long is it in the air? Assume it comes back to the same location and air resistance is negligible. 2) A car traveling at...
-
1. Suppose the Cobb-Douglas production function in an economy is Y=KL", where K is the amount of capital and Z is the amount of labor. The economy begins with 60 units of capital and 100 units of...
-
Consider an economy populated by Farmer Freddy, Baker Betty, and a bunch of consumers. Freddy and Betty each own their respective businesses, so they keep all profits. Freddy hires labor and grows...
-
Person should choose schooling such that the marginal rate of return equals the discount rate . Suppose the discount rate is r but the person stops schooling at s' and starts working for wage w'. If...
-
Explain the concept of higher-order functions in functional programming. How do you use them to create more concise and expressive code, and what are the benefits of immutability in this paradigm?
-
Journalize these 1. The Pink Boutique Co. provided clothing services to Forever 21 worth $7000 that were not billed on October 31. 2. On November 10, Sanareah receives cash of $7,000 for services...
-
One hundred pounds of water at atmospheric pressure are heated from 60F to 200F. What is the enthalpy change? The internal energy change? Why is the difference between the internal energy change and...
-
Data from Tables 6.3 and 6.4 are needed for this problem. (a) Estimate the lattice energy of CsCl if the CsCl internuclear distance is 356.6 pm. (b) Now consider a polymorph of CsCl that crystallizes...
-
Measures taken to control atmospheric pollution include (a) Scrubbing industrial waste gases to remove SO 2 , (b) Reduction of NO in motor vehicle emissions. Explain how these are achieved and write...
-
A crucial component of a dye-sensitized solar cell (DSC) is the sensitizer. The latter captures photons which are converted to electric current in the cell. A typical dye is cis-[Ru(L) 2 (NCSN) 2 ]...
-
Which is NOT a class of SoS design variables? a. Composition b.Configuration c. Control d . Communication
-
Design a system hierarchy forareal- world System of Systems problem. Perform background research on the different levels of system hierarchies and decide on an application of interest .Discuss why...
-
What are the ROPE table categories? a. Research, Operations, Project, Economics b.Resources, Operations, Politics, Environment c. Resources, Operations, Policy, Economics d. Reusability, Operations...

Study smarter with the SolutionInn App


