On October 1, 2024, the Allegheny Corporation purchased equipment for $203,000. The estimated service life of...
Fantastic news! We've Found the answer you've been seeking!
Question:
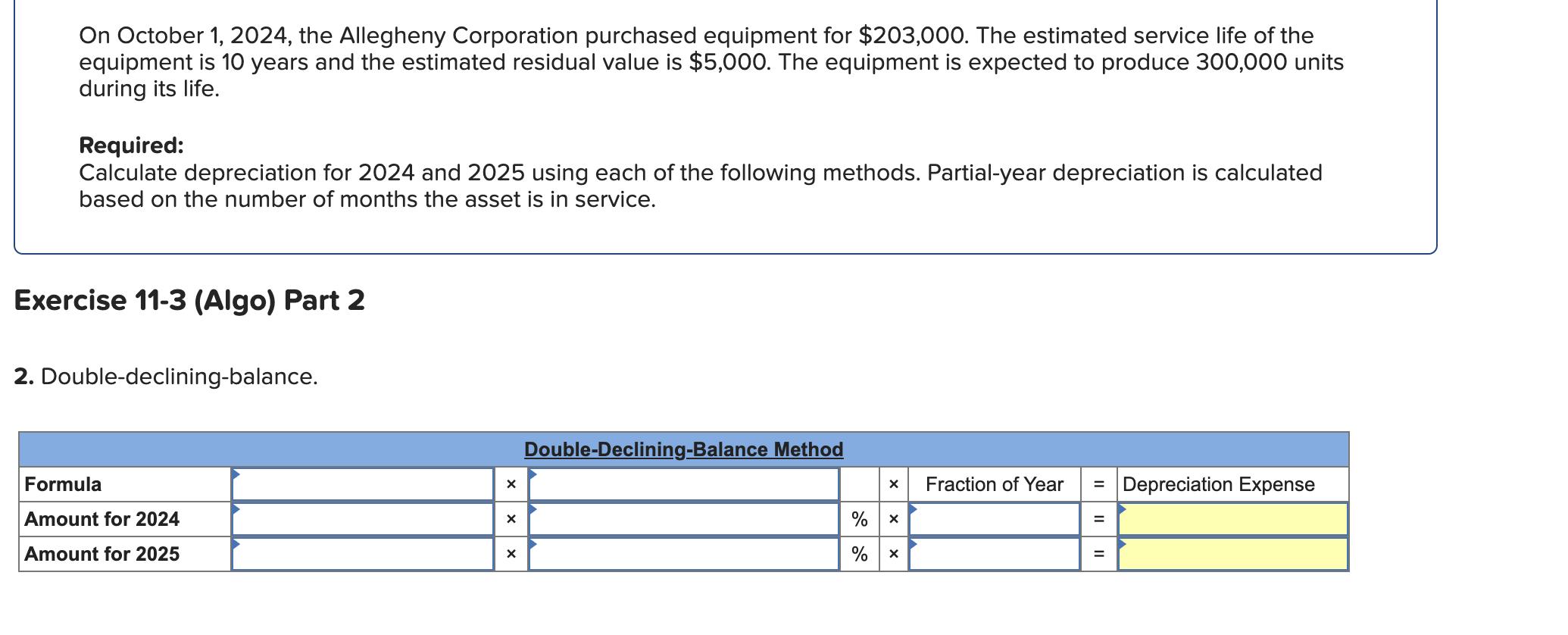
Transcribed Image Text:
On October 1, 2024, the Allegheny Corporation purchased equipment for $203,000. The estimated service life of the equipment is 10 years and the estimated residual value is $5,000. The equipment is expected to produce 300,000 units during its life. Required: Calculate depreciation for 2024 and 2025 using each of the following methods. Partial-year depreciation is calculated based on the number of months the asset is in service. Exercise 11-3 (Algo) Part 2 2. Double-declining-balance. Formula Amount for 2024 Amount for 2025 Double-Declining-Balance Method % % x Fraction of Year II = Depreciation Expense On October 1, 2024, the Allegheny Corporation purchased equipment for $203,000. The estimated service life of the equipment is 10 years and the estimated residual value is $5,000. The equipment is expected to produce 300,000 units during its life. Required: Calculate depreciation for 2024 and 2025 using each of the following methods. Partial-year depreciation is calculated based on the number of months the asset is in service. Exercise 11-3 (Algo) Part 2 2. Double-declining-balance. Formula Amount for 2024 Amount for 2025 Double-Declining-Balance Method % % x Fraction of Year II = Depreciation Expense
Expert Answer:
Posted Date:
Students also viewed these accounting questions
-
How preventive and responsive supply chain enablers lead to a more effective risk management.
-
18. Equal volumes of two solutions containing 3.65g of HCl and 4.0 g of NaOH respectively are mixed. The pli of the mixture is 2) <7 3) >7 19. When Imi of 0.IN HCIIS added to 1 litre of a solution of...
-
Consider the situation in problem 2-79. Three consumers are chosen randomly from among a group of potential buyers of the high-performance automobile. What is the probability that all three of them...
-
Hass Company estimates that 360,000 direct labor hours will be worked during the coming year, 2011, in the Packaging Department. On this basis, the budgeted manufacturing overhead cost data, shown on...
-
Clarkson Company is a large multi-division firm with several plants in each division. A comprehensive budgeting system is used for planning operations and measuring performance. The annual budgeting...
-
Pueblo Enterprises is considering investing in either of two mutually exclusive projects, X and Y. Project X requires an initial investment of $30,000; project Y requires $40,000. Each projects cash...
-
Consider the follwing characteristics of the photoelectric effect: The generation of photoelectrons The existence of a threshold frequency The photoelectric current increases with increasing light...
-
Question 1 [25 Marks] Research planning is subject to various influences that exist in the research context. The associated rescarch paradigms, approaches, methodologies and designs that make up the...
-
PACIFIC HEALTHCARE IS an investor-owned hospital chain that owns and operates nine hospitals in Washington, Oregon, and Northern California Marcia Long, a recent graduate of a prominent health...
-
On January 1, 2024, the Mason Manufacturing Company began construction of a building to be used as its office headquarters. The building was completed on September 30, 2025. Expenditures on the...
-
A ball is dropped from a high building. Using the approximate value of g = 1 0 m / s 2 , determine its change in speed from 1 s to 1 1 s in flight.
-
A certain laboratory experiment requires an aluminum wire of length of 3 2 . 0 m and a resistance of 2 . 5 0 V at 2 0 . 0 \ deg C . What diameter wire must be used?
-
The historical development of the theory (what lead to the theory) 2. Key ideas/main ideas of the theory 3. Critically evaluate the effectiveness of the theory. on the journal article by Kempster,...
-
What would be the change in pressure in a sealed 10.0 L vessel due to the formation of N2 gas when the ammonium nitrite in 2.00 L of 1.40 M NH4NO2 decomposes at 25.0C?
-
How do network effects help Facebook fend off smaller social-networking rivals? Could an online retailer doing half as much business compete on an equal footing with Amazon in terms of costs? Explain.
-
What control procedures would you recommend in each of the following situations? 1. A concession company has one employee who sells sunscreen, T-shirts, and sunglasses at the beach.Each day, the...
-
Del Gato Clinic deposits all cash receipts on the day they are received and it makes all cash payments by check. At the close of business on June 30, 2008, its Cash account shows an \($11,589\) debit...
-
Read the article No Accounting for Being Disorganized in the March 10, 2004, issue of BusinessWeek. (The books Website provides a free link.) Required 1. Why does Steven Cohen, the author of the...

Study smarter with the SolutionInn App


