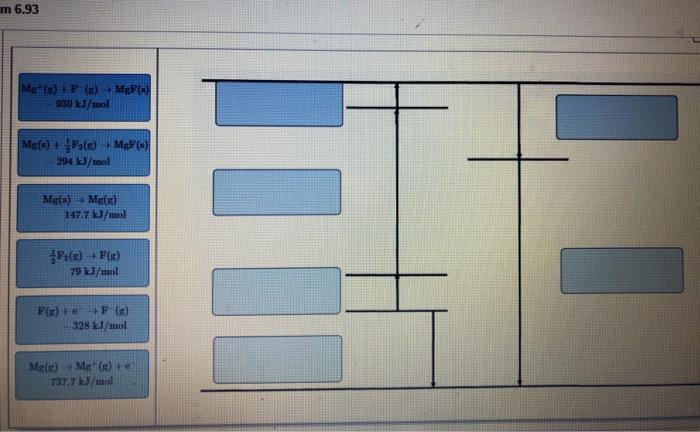
Part A Draw Born-Haber cycles for the formation of both MgF. Drag the appropriate labels to...
Fantastic news! We've Found the answer you've been seeking!
Question:

Transcribed Image Text:
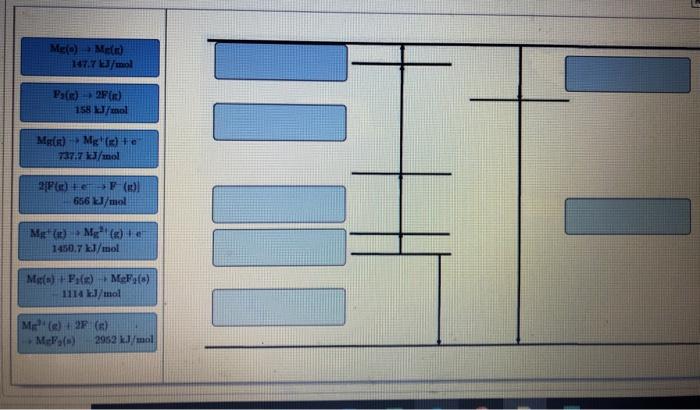
Part A Draw Born-Haber cycles for the formation of both MgF. Drag the appropriate labels to their respective targets. m 6.93 Mg (n)+F (n)→ MgF(x) 930 kJ/mol Mg(s) + F(x) +MgF(s) 204 kJ/mol Mg(s) Mg(g) 147.7 kJ/mol (2)→ F(x) 79 kJ/mol F(g) + +F (n) 328 kJ/mol Mg(s) Mg(s) +e 737.7 kJ/mol 5 II Y Part B Draw Born-Haber cycles for the formation of MgF₂. Drag the appropriate labels to their respective targets. Mg(0) Me(n) 147.7 kJ/mol Par) 2F(x) 158 kJ/mol Mg() Mg(x) to 737.7 kJ/mol 2/P(x) + e SF ()) 656 kJ/mol M Mg()+ 1450.7kJ/mol Mg() + F(x) MeFy(s) 1114 kJ/mol M (2) 2F (1) MePa(s) 2952 kJ/mol III Part A Draw Born-Haber cycles for the formation of both MgF. Drag the appropriate labels to their respective targets. m 6.93 Mg (n)+F (n)→ MgF(x) 930 kJ/mol Mg(s) + F(x) +MgF(s) 204 kJ/mol Mg(s) Mg(g) 147.7 kJ/mol (2)→ F(x) 79 kJ/mol F(g) + +F (n) 328 kJ/mol Mg(s) Mg(s) +e 737.7 kJ/mol 5 II Y Part B Draw Born-Haber cycles for the formation of MgF₂. Drag the appropriate labels to their respective targets. Mg(0) Me(n) 147.7 kJ/mol Par) 2F(x) 158 kJ/mol Mg() Mg(x) to 737.7 kJ/mol 2/P(x) + e SF ()) 656 kJ/mol M Mg()+ 1450.7kJ/mol Mg() + F(x) MeFy(s) 1114 kJ/mol M (2) 2F (1) MePa(s) 2952 kJ/mol III
Expert Answer:
Answer rating: 100% (QA)
BornHaber Cycle for MgF2 Formation Part A In the BornHaber cycle for the formatio... View the full answer

Related Book For 

Posted Date:
Students also viewed these accounting questions
-
The equilibrium constant (KP) for the formation of the air pollutant nitric oxide (NO) in an automobile engine at 530°C is 2.9 Ã 10-11: (a) Calculate the partial pressure of NO under these...
-
A proposed mechanism for the formation of N 2 O 5 (g) from NO 2 (g) and O 3 (g) is Determine the rate law expression for the production of N 2 O 5 (g) given this mechanism. NO,(g) + O3(g)1...
-
The standard free energy change for the formation of H 2 (g) + O 2 (g) from H 2 O is G = + 237.13 kJ. The reactions are Calculate the standard voltage (E) needed to decompose water into its elements...
-
Bruno Corp. has decided to expand its operations. The bookkeeper recently completed the following statement of financial position in order to obtain additional funds for expansion: Instructions (a)...
-
The analyst in Exercise 28 realized the data were in need of transformation because of the nonlinearity between the variables. Economists commonly take the logarithm of these variables to make the...
-
The table that follows lists four pairs of initial and final angular velocities for a rotating fan blade. The elapsed time for each of the four pairs of angular velocities is 4.0 s. For each of the...
-
For the multiple linear regression model, show that $S S_{\mathrm{R}}(\boldsymbol{\beta})=\mathbf{y}^{\prime} \mathbf{H y}$.
-
Prepare a performance report for the dining room of Leonardos Italian Cafe´ for the month of February 2011, using the followingdata: Budgeted Data: Dining room wages.. Laundry and housekeeping....
-
A $1,000 par value bond that pays interest annually just paid $106 in interest. What is the coupon rate? Q2) A 09.60% coupon, 21-year annual bond is priced at $986. What is the current yield for...
-
Calculate the NPV of the proposed overhaul of the Vital Spark, with and without the new engine and control system. To do the calculation, you will have to prepare a spreadsheet table showing all...
-
Treatment of compound D with LIAIH4 followed by H20 forms compound E. D shows a molecular ion in its mass spectrum at m/z 71 and IR absorptions at 3600 - 3200 and 2263 cm-1. E shows a molecular ion...
-
If an interest rate is currently 6% and a lending institution announces a 25 basis point increase, what percentage increase does this represent? Calculate the straight-line and sum-of-years-digits...
-
For every buyer of, say, a call option, there must of course also be a seller. Why would someone sell a call option on some shares he or she already owned? How would this be different than buying a...
-
The Clinton administration proposed in the spring of 1993 to shorten the average maturity of the federal debt to reduce interest outlays in the budget. How would the administration do this, and what...
-
A portfolio has 70% investment weight in V, and the rest 30% investment weight in W. V has the standard deviation of 10%; W has the standard deviation of 20%. V and Whave the correlation coefficient...
-
The local paper delivery person has to purchase daily newspapers prior to reselling them. Newspapers cost $0.75 each, and can be sold for $1.25 each. At the end of each day, the paper deliverer can...
-
Analyze China's International trade in the last one year and also assess its impact on the balance of payments and exchange rate of its economy. Guidelines: . Discuss in brief the trade variations...
-
1. Using the information from Problem 16-4B, prepare a statement of cash flows for Lim Garden Supplies Inc. using the direct method of presenting cash flows from operating activities. 2. How does Lim...
-
The second ionization energy of magnesium is only about twice as great as the first, but the third ionization energy is 10 times as great. Why does it take so much more energy to remove the third...
-
At 25C, DG for the process H2O(l) H2O(g) is 8.6 kJ/mol. Calculate the vapor pressure of water at this temperature.
-
The following are common household compounds: table salt (NaCl), table sugar (sucrose), vinegar (contains acetic acid), baking soda (NaHCO3), washing soda (Na2CO3? 10H2O), boric acid (H3BO3, used in...
-
Henry Ford, founder of Ford Motor Company, is quoted as saying that customers could choose a car in any color as long as it was black. Things have come a long way since that timewhen customization...
-
Not long ago, New England Confectionery Company, or Necco for short, marked the production of its one trillionth candy wafer. The humble roots of Necco, the country's oldest continuously operating...
-
What is utility?

Study smarter with the SolutionInn App


