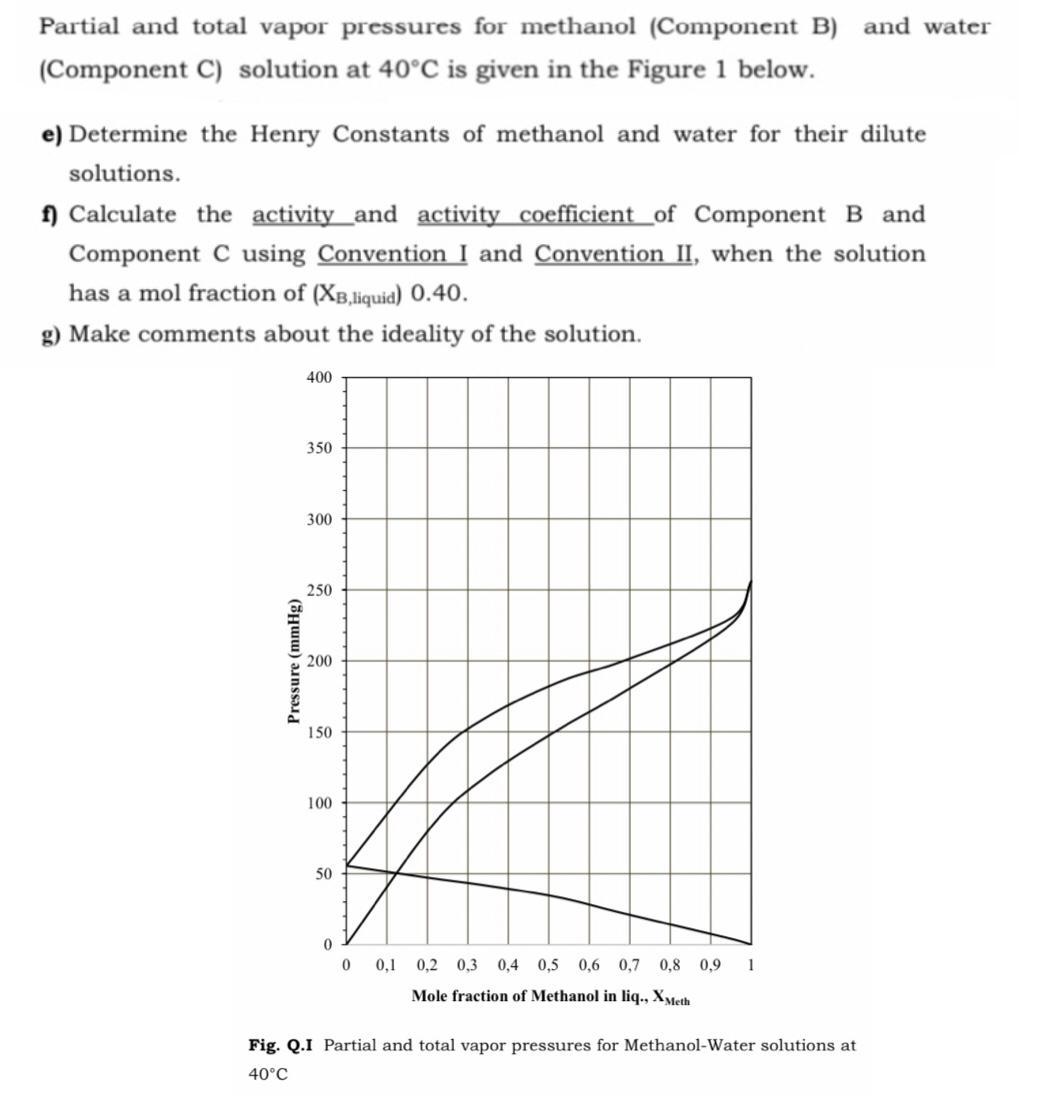
Partial and total vapor pressures for methanol (Component B) and water (Component C) solution at 40C...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
Partial and total vapor pressures for methanol (Component B) and water (Component C) solution at 40°C is given in the Figure 1 below. e) Determine the Henry Constants of methanol and water for their dilute solutions. 1 Calculate the activity _and activity coefficient_of Component B and Component C using Convention I and Convention II, when the solution has a mol fraction of (XB,iquid) 0.40. g) Make comments about the ideality of the solution. 400 350 300 250 200 150 100 50 0,1 0,2 0,3 0,4 0,5 0,6 0,7 0,8 0,9 1 Mole fraction of Methanol in liq., Xyeth Fig. Q.I Partial and total vapor pressures for Methanol-Water solutions at 40°C Pressure (mmHg) Partial and total vapor pressures for methanol (Component B) and water (Component C) solution at 40°C is given in the Figure 1 below. e) Determine the Henry Constants of methanol and water for their dilute solutions. 1 Calculate the activity _and activity coefficient_of Component B and Component C using Convention I and Convention II, when the solution has a mol fraction of (XB,iquid) 0.40. g) Make comments about the ideality of the solution. 400 350 300 250 200 150 100 50 0,1 0,2 0,3 0,4 0,5 0,6 0,7 0,8 0,9 1 Mole fraction of Methanol in liq., Xyeth Fig. Q.I Partial and total vapor pressures for Methanol-Water solutions at 40°C Pressure (mmHg)
Expert Answer:

Related Book For 

Posted Date:
Students also viewed these chemical engineering questions
-
The bolted joint given in the figure is being applied on by the load P which is fluctuating between 0 and 2x10* N. Both of the clamped parts are made of steel (E = 207 GPa). The bolt is M12x1.75, IsO...
-
Determine the quadratic function whose graph is given in the figure. YA 50 (0, 24) -2 8 -10F Vertex: (4, -8)
-
Starting from a calculus textbook definition of radius of curvature and the equation of an ellipse, derive the following formula representing the meridian radius of curvature: M = a(1-e) (1 e sin )/...
-
Access various employment Web sites (for example, www.monster.com and www.dice.com ) and find several job descriptions for a database administrator. Are the job descriptions similar? What are the...
-
Suppose again that the examination considered in Exercise 13 is taken by five students. Determine the probability that no two students will complete the examination within 10 minutes of each other.
-
Table 19-2 indicates that the short-run price elasticity of demand for tires is 0.9. If an increase in the price of petroleum (used in producing tires) causes the market prices of tires to rise from...
-
Electrons are emitted when a metal is illuminated by light with a wavelength less than \(388 \mathrm{~nm}\) but for no greater wavelength. What is the metal's work function?
-
The New City College reported deferred revenues of $607,000 as of July 1, 2011, the first day of its fiscal year. Record the following transactions related to student tuition and fees and related...
-
Your employer is trying to select from a list of possible capital projects. The projects, along with their cost and benefits, are listed below. The capital budget available is $1 million. In addition...
-
Fresno Industries Inc. manufactures and sells high-quality camping tents. The company began operations on January 1 and operated at 100% of capacity (150,000 units) during the first month, creating...
-
Which of the follow are contributors to the extraordinary cost of the US healthcare systems compared to other OECD countries [select all that apply]? We spend a lot on non-durable medical care and...
-
Ken Barker received a $10,000 scholarship to study undergraduate engineering at Rainbow University for the current academic year. Ken received the $10,000 in cash, and he used the money as follows:...
-
Why do I have to know how a bill becomes a law? Why is common law important? Discuss the significance of common laws for the healthcare professional and the differentiate between common law and...
-
What are the challenges and controversies surrounding the use of morphological characters versus molecular data in taxonomic classification, and how can these approaches be effectively integrated to...
-
On 1 / 1 / 2 3 , Jeckle Technologies Inc. paid $ 4 , 7 9 2 , 0 8 5 to acquire $ 5 , 0 0 0 , 0 0 0 in bonds that mature in 5 years. The bonds pay interest semi - annually at 6 % per annum on 6 / 3 0...
-
1, Do all problems stem back to one or more root causes, or is it possible that some causes are simply active and exist without roots? 2, Explain Reason's "Swiss Cheese" model of defensive layers to...
-
Your company bought a new distribution facility 3 years ago for $9 million. Your company currently has two options: (A) Sell the facility today for $7 million (before taxes). (B) Alternatively, if...
-
After Theorem 1.5 we note that multiplying a row by 0 is not allowed because that could change a solution set. Give an example of a system with solution set S0 where after multiplying a row by 0 the...
-
The polar coordinates of a point are given. Find the rectangular coordinates of the point. (-2, 3 /4)
-
Find the exact value of expression. Do not use a calculator. 3 sin 45 4 tan 6.
-
The navigator of a ship at sea spots two lighthouses that she knows to be 3 miles apart along a straight seashore. She determines that the angles formed between two line-of-sight observations of the...
-
A change in chromosome structure that does not involve a change in the total amount of genetic material is a. a deletion. b. a duplication. c. an inversion. d. none of the above.
-
The term endopolyploidy refers to the phenomenon of having a. too many chromosomes. b. extra chromosomes inside the cell nucleus. c. extra sets of chromosomes in certain cells of the body. d. extra...
-
During transduction involving a P1 phage, a. any small fragment of the bacterial chromosome may be transferred to another bacterium by a new phage. b. only a specific fragment of DNA may be...

Study smarter with the SolutionInn App


