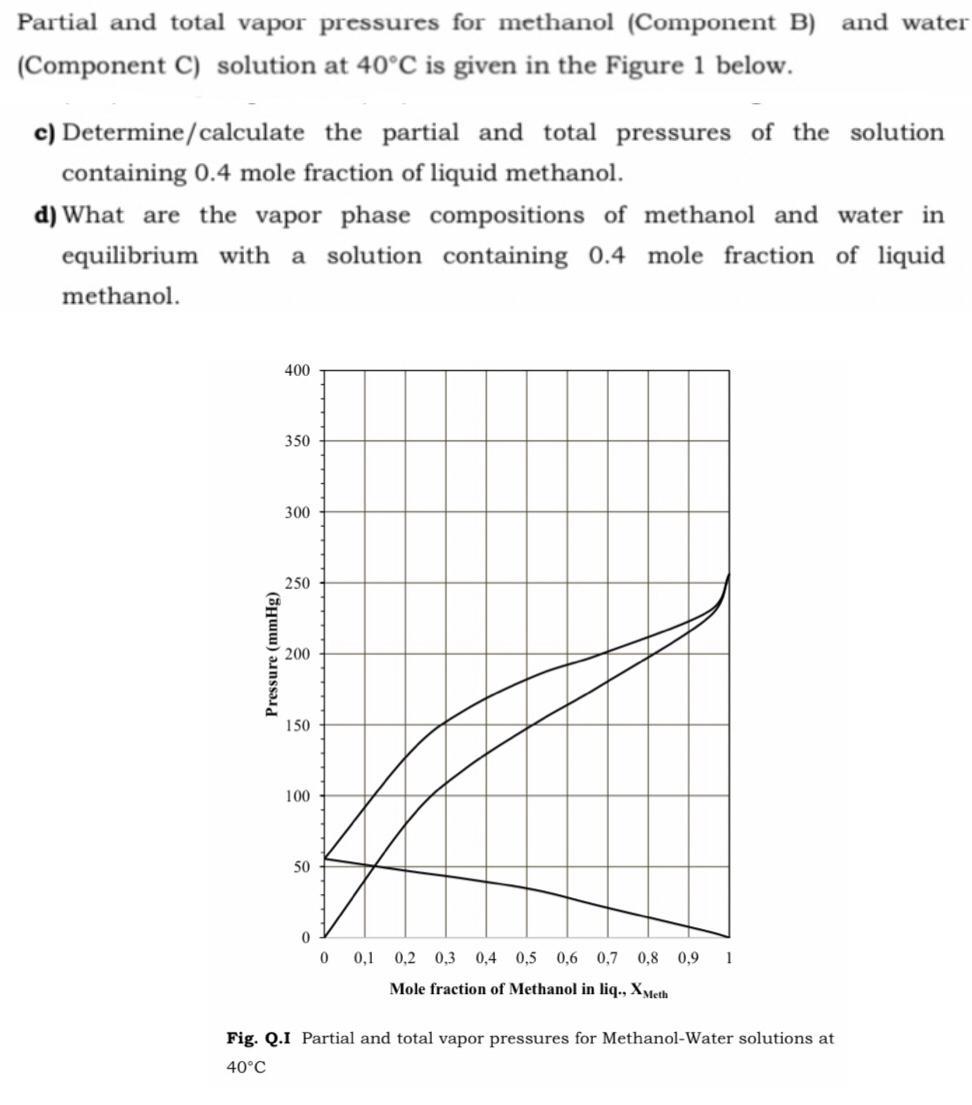
Partial and total vapor pressures for methanol (Component B) and water (Component C) solution at 40C...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
Partial and total vapor pressures for methanol (Component B) and water (Component C) solution at 40°C is given in the Figure 1 below. c) Determine/calculate the partial and total pressures of the solution containing 0.4 mole fraction of liquid methanol. d) What are the vapor phase compositions of methanol and water in equilibrium with a solution containing 0.4 mole fraction of liquid methanol. 400 350 300 250 200 150 100 50 0,1 0,2 0,3 0,4 0,5 0,6 0,7 0,8 0,9 1 Mole fraction of Methanol in liq., XMeth Fig. Q.I Partial and total vapor pressures for Methanol-Water solutions at 40°C Pressure (mmHg) Partial and total vapor pressures for methanol (Component B) and water (Component C) solution at 40°C is given in the Figure 1 below. c) Determine/calculate the partial and total pressures of the solution containing 0.4 mole fraction of liquid methanol. d) What are the vapor phase compositions of methanol and water in equilibrium with a solution containing 0.4 mole fraction of liquid methanol. 400 350 300 250 200 150 100 50 0,1 0,2 0,3 0,4 0,5 0,6 0,7 0,8 0,9 1 Mole fraction of Methanol in liq., XMeth Fig. Q.I Partial and total vapor pressures for Methanol-Water solutions at 40°C Pressure (mmHg)
Expert Answer:

Related Book For 

Posted Date:
Students also viewed these chemical engineering questions
-
The bolted joint given in the figure is being applied on by the load P which is fluctuating between 0 and 2x10* N. Both of the clamped parts are made of steel (E = 207 GPa). The bolt is M12x1.75, IsO...
-
Determine the quadratic function whose graph is given in the figure. YA 50 (0, 24) -2 8 -10F Vertex: (4, -8)
-
Starting from a calculus textbook definition of radius of curvature and the equation of an ellipse, derive the following formula representing the meridian radius of curvature: M = a(1-e) (1 e sin )/...
-
Various streptococci and lactobacilli were traditionally grouped together as lactic acid bacteria because of their characteristic fermentation. Most of them were found to have a DNA Guanine plus...
-
It is said that a random variable X has the Pareto distribution with parameters x0 and (x0 > 0 and > 0) if X has a continuous distribution for which the p.d.f. f (x|x0, ) is as follows: Show that...
-
1. Would a U6 version of the natural unemployment rate likely be higher or lower than the traditional natural unemployment rate? Explain your reasoning? 2. Why would using the U6 unemployment rate...
-
Metal surfaces on spacecraft in bright sunlight develop a net electric charge. Do they develop a negative or a positive charge? Explain.
-
Armstrong Faber produces a standard number-two pencil called Ultra-Lite. Since Chuck Armstrong started Armstrong Faber, sales have grown steadily. With the increase in the price of wood products,...
-
An analyst has calculated the CVA for a 4-year 6% $100 annual coupon paying bond to be 4.256. If the risk-free rate was 2%, what would be the fair value of the risky bond? Give your answer correct to...
-
Whats the difference between a real resource and a virtual resource?
-
You notice that no one has created a method to apply overhead to the products, and they are currently being sold without this value added to them. Currently, all overhead is considered an...
-
Prepare a classified Balance Sheet, dated 12-31-18, for these 3 situations: Situation 1 12-31-18 Assets: Cash . 183,000 Accounts Rec. ..... 274,000 Inventory .... 175,000 Equipment, Net of Accum....
-
With dealing with intellectual property, there are two ways it can be enforced, being common law and statutory law. For example with patents, what would be the "punishment" or enforcement I should...
-
Why do problems at work stay undetected for so long? Does anyone like to be wrong? How do you react when someone informs you of a mistake? Can you think of any specific examples and reactions that...
-
Suppose you run a business making pizzas. Over a year, you pay $18,000 for the ingredients, equipment, etc. that you need to make the pizzas, and your total revenue from selling pizza is $60,000. Had...
-
How do mutualistic interactions contribute to the resilience of agricultural systems, and what strategies can be employed to enhance the sustainability and productivity of agroecosystems through the...
-
A negatively charged particle is moved into the field generated by a positive charge. (a) Draw the field lines around both charges. (b) The negative charge is 52 nC and the positive charge is 27 nC....
-
For liquid water the isothermal compressibility is given by; where r and b are functions of temperature only. If 1 kg of water is compressed isothermally and reversibly from I to 500 bar at 60(C. how...
-
Find the exact value of expression. tan 195
-
In problem, the function f is one-to-one. Find its inverse and check your answer. Graph f, f -1 , and y = x on the same coordinate axes. f(x) = x 2 + 9 x 0
-
In problem, solve each equation. log 5 x = 3
-
If a tRNA has an anticodon with the sequence 3-GAC-5, which amino acid does it carry? a. Aspartic acid c. Leucine b. Valine d. Glutamine
-
Lets suppose a researcher mixed together nucleotides with the following percentages of bases: 30% G, 30% C, and 40% A. If RNA was made via polynucleotide phosphorylase, what percentage of the codons...
-
An inborn error of metabolism is caused by a. a mutation in a gene that causes an enzyme to be inactive. b. a mutation in a gene that occurs in somatic cells. c. the consumption of foods that disrupt...

Study smarter with the SolutionInn App


