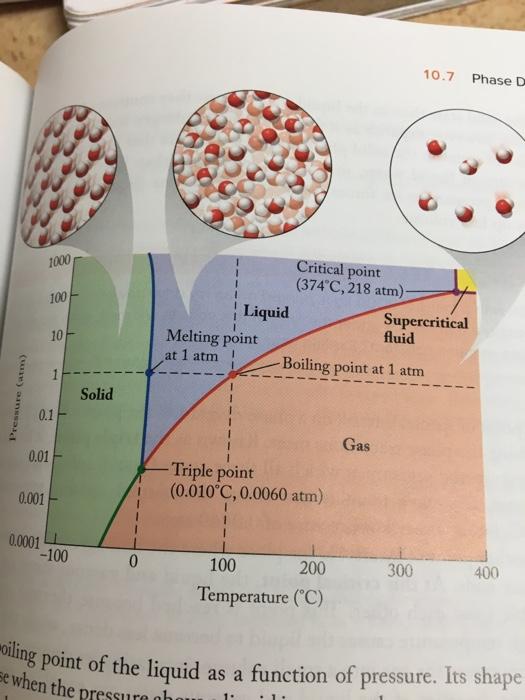
Predict the phase of water that exists under the following conditions. a. 2 atm of pressure...
Fantastic news! We've Found the answer you've been seeking!
Question:
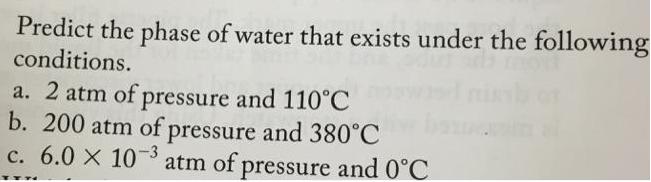
Transcribed Image Text:
Predict the phase of water that exists under the following conditions. a. 2 atm of pressure and 110°C b. 200 atm of pressure and 380°C c. 6.0 X 103 atm of pressure and 0°C oiling point of the liquid as a function of p se when the pressure obo 10.7 Phase D Critical point (374 C, 218 atm)- 1000 100 Liquid Melting point Supercritical fluid 10 at 1 atm Boiling point at 1 atm Solid 0.1- Gas 0.01 Triple point (0.010°C, 0.0060 atm) 0.001 0.0001 -100 0. 100 200 300 400 Temperature ("C) ling point of the liquid as a function of pressure. Its shape 1. 0 Intermolecular Forces 1000 Diethyl ether (CH,CH,OCH,CH Bromine (Br) Normal boiling point 1 atm) rapor pressure quids shows that increasing at which the the normal 900 - Ethanol (CH;CH,OH) - 800 700 600 (760 torr = - Water (H,0) - 500 400 300 Octane [CH;(CH),CH Ethylene glycol (HOCH,CH,OH) - 200 100 -100 -50 0 50 100 150 200 250 300 350 400 Temperature ("C) Vapor pressure (mmHg) Predict the phase of water that exists under the following conditions. a. 2 atm of pressure and 110°C b. 200 atm of pressure and 380°C c. 6.0 X 103 atm of pressure and 0°C oiling point of the liquid as a function of p se when the pressure obo 10.7 Phase D Critical point (374 C, 218 atm)- 1000 100 Liquid Melting point Supercritical fluid 10 at 1 atm Boiling point at 1 atm Solid 0.1- Gas 0.01 Triple point (0.010°C, 0.0060 atm) 0.001 0.0001 -100 0. 100 200 300 400 Temperature ("C) ling point of the liquid as a function of pressure. Its shape 1. 0 Intermolecular Forces 1000 Diethyl ether (CH,CH,OCH,CH Bromine (Br) Normal boiling point 1 atm) rapor pressure quids shows that increasing at which the the normal 900 - Ethanol (CH;CH,OH) - 800 700 600 (760 torr = - Water (H,0) - 500 400 300 Octane [CH;(CH),CH Ethylene glycol (HOCH,CH,OH) - 200 100 -100 -50 0 50 100 150 200 250 300 350 400 Temperature ("C) Vapor pressure (mmHg)
Expert Answer:
Answer rating: 100% (QA)
a According to the phase diagram at 2 atm pressure and 110 degrees ... View the full answer

Related Book For 

Thermodynamics An Engineering Approach
ISBN: 978-0073398174
8th edition
Authors: Yunus A. Cengel, Michael A. Boles
Posted Date:
Students also viewed these physics questions
-
Estimating Cost of Capital for Netflix company a. Compute the market value of all of the companys equity. b. Compute the market value of the companys interest-bearing debt, using the interest...
-
Diethyl ether has a boiling point of 34.5°C, and 1-butanol has a boiling point of 117°C: Both of these compounds have the same numbers and types of atoms. Explain the difference in their...
-
Water is boiling at 1 atm pressure in a stainless steel pan on an electric range. It is observed that 2 kg of liquid water evaporates in 30 min. The rate of heat transfer to the water is (a) 2.51 kW...
-
Case 2: First Year on the Job Joe finally received that job offer in the mail. He worked part-time for First Cirrus Bank during the spring semester and the work experience had paid off. Beginning on...
-
If global warming caused a 2-meter rise in worldwide sea level, what would happen to the price of land? How would that result affect your calculation of the benefits of slowing climate change?
-
Identify at least two plausible service departments in your industry* (not including HR, IT or maintenance). Describe the departments, the services they provide to the producing departments or to...
-
Explain with a neat sketch the working of fast and loose pulley.
-
Multiple Choice Questions The following questions deal with confirmation of accounts receivable. Choose the best response. a. The negative form of accounts receivable confirmation request is useful...
-
Your text talks about Therapeutic Use Exemptions (TUE) and how they play a role in modern drug testing. You've also read a portion of the Major League Baseball Joint Drug Prevention and Treatment...
-
Upsidedown Cake Company produces dessert products for sale in grocery stores, but it also has a retail location. At the end of 2018, the company had $349,000 in accounts receivable before netting out...
-
The weight W of the building is 200 kips and the building is set into free vibration by releasing it (at time t = 0) from a displacement of 1.20 in. If the maximum displacement on the return swing is...
-
What is different about an EVA-based contingent budgeting and control system?
-
What is variance analysis? What role does it play in the control process?
-
Would you expect the bid-ask percentage spread to be different for the Indonesian rupiah versus the U.S. dollar than for the Japanese yen?
-
What is the difference between territorial and worldwide tax regimes?
-
State the Law of One Price. Why does it hold in the foreign exchange market?
-
All of the following items would qualify as hedging instruments, except Multiple choice question. futures contracts. interest rate swaps. insurance contracts. call options.
-
Calculate the number of neutrons of 239Pu.
-
A person gets into an elevator at the lobby level of a hotel together with his 30-kg suitcase, and gets out at the 10th floor 35 m above. Determine the amount of energy consumed by the motor of the...
-
Using EES (or other) software, investigate the effect of the condenser pressure on the performance of a simple ideal Rankine cycle. Turbine inlet conditions of steam are maintained constant at 10 MPa...
-
Determine the rate of net heat gain (or loss) through a 9-ft-high, 15-ft-wide, fixed 18 -in single-glass window with aluminum frames on the west wall at 3 PM solar time during a typical day in...
-
Temperature is an intensive property. State True (T) or False (F)
-
Kinetic energy is an extensive property. State True (T) or False (F)
-
A nozzle represents an open system. State True (T) or False (F)

Study smarter with the SolutionInn App


