n ? Every radioactive isotope decays at a certain rate It is never known when a given
Fantastic news! We've Found the answer you've been seeking!
Question:
 n
n
Transcribed Image Text:
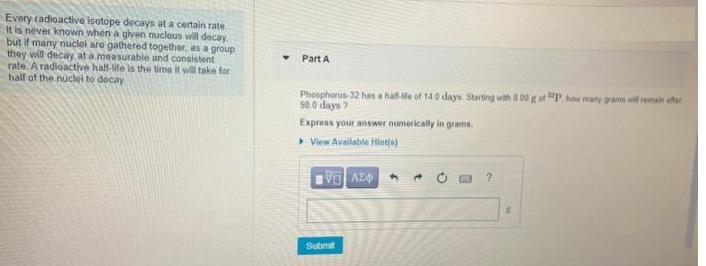
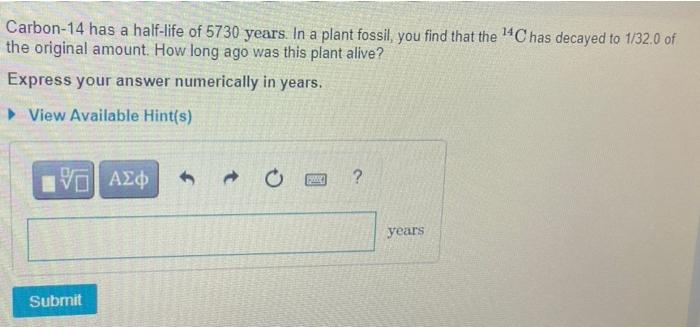
Every radioactive isotope decays at a certain rate It is never known when a given nucleus will decay but if many nuclei are gathered together, as a group they will decay at a measurable and consistent rate. A radioactive half-life is the time it will take for half of the nuclei to decay Part A Phosphorus-32 has a half-le of 140 days Starting with 800 g of P. how many grams will remain after 98.0 days 7 Express your answer numerically in grams. >View Available Hint(s) VAZO Submit 4 A ? C Carbon-14 has a half-life of 5730 years. In a plant fossil, you find that the ¹4C has decayed to 1/32.0 of the original amount. How long ago was this plant alive? Express your answer numerically in years. View Available Hint(s) VE ΑΣΦ Submit PAC years Every radioactive isotope decays at a certain rate It is never known when a given nucleus will decay but if many nuclei are gathered together, as a group they will decay at a measurable and consistent rate. A radioactive half-life is the time it will take for half of the nuclei to decay Part A Phosphorus-32 has a half-le of 140 days Starting with 800 g of P. how many grams will remain after 98.0 days 7 Express your answer numerically in grams. >View Available Hint(s) VAZO Submit 4 A ? C Carbon-14 has a half-life of 5730 years. In a plant fossil, you find that the ¹4C has decayed to 1/32.0 of the original amount. How long ago was this plant alive? Express your answer numerically in years. View Available Hint(s) VE ΑΣΦ Submit PAC years
Expert Answer:
Answer rating: 100% (QA)
Part A Half life 14 days use relation between rate constant and half life of 1st order re... View the full answer

Related Book For 

Elementary Principles of Chemical Processes
ISBN: 978-0471720638
3rd Edition
Authors: Richard M. Felder, Ronald W. Rousseau
Posted Date:
Students also viewed these physics questions
-
N balls labeled 1 through N are placed in Box 1 while a Box 2 is initially empty. At each time instant, one of the N balls is chosen (with equally probability) and moved to the other box. Let X [k]...
-
N sources of current with different emf's are connected as shown in Fig. 3.40. The emf's of the sources are proportional to their internal resistances, i.e. ε = aR, where a is an assigned...
-
N = the set of natural numbers, W = the set of whole numbers, Z = the set of integers, Q = the set of rational numbers, I = the set of irrational numbers, and R = the set of real numbers. Classify...
-
You have three light bulbs; bulb A has a resistance of 240 , bulb B has a resistance of 192 , and bulb C has a resistance of 144 . Each of these bulbs is used for the same amount of time in a setup...
-
a. Create a logistic regression model using Gender and Age to estimate the probability of survival. Create a plot of the estimated probability of survival using Age as the explanatory variable and...
-
The Fagersta Steelworks currently is working two mines to obtain its iron ore. This iron ore is shipped to either of two storage facilities. When needed, it then is shipped on to the company's steel...
-
The femoral artery is the large artery that carries blood to the leg. A person's femoral artery has an inner diameter of \(1.0 \mathrm{~cm}\). What is the resistance of a 20-cm-long column of blood...
-
Texcar Co. is considering the following alternative financing plans: Income tax is estimated at 40% of income. Determine the earnings per share of common stock, assuming income before bond interest...
-
Consider a Poisson GLM with the log link and linear predictor of the form Ni = B1 + B2ai, E {1,...n}, = ni where ai is the value of a factor predictor with two levels, such that ai 1 for {1,... Ni}...
-
A. Ray and Maria Gomez have been married for 3 years. Ray is a propane salesman for Palm Oil Corporation and Maria works as a city clerk for the City of McAllen. Rays birthdate is February 21, 1990...
-
2. An electronic component schematically shown in Figure 2 is made up of two different materials with thermal conductivities K = 50W/mK and K = 10W/mK, densities p=1500kg/m and P = 2000kg/m, and...
-
H Bilberry purchased a machine on 31 July 2021, to be depreciated by the straight line method at 15% p.a., which cost $26 312 ($23 920 + $2392 GST). Installation and commissioning was completed on 31...
-
Prepare the accounts receivable and accounts payable control accounts of J Wright for November 2022. Accounts payable control balance 31 October 2022 Accounts receivable control balance 31 October...
-
A business involved with long-distance transport bought a new truck designed for haulage between all capital cities throughout the country. The motor vehicle was purchased on credit on 31 July 2021...
-
Plant and equipment was purchased on credit on 1 September 2021 for $85 415 ($77 650 + $7765 GST). Installation was finalised and commissioned on 31 October 2021 at a cost of $18 821 ($17 110 + $1711...
-
As part of a major expansion, the business bought on credit equipment at a cost including installation of $77 968 ($70 880 + $7088 GST). Installation and commissioning were completed on 1 February...
-
Alliance Company Limited is constructing a Plant that is financed with $10 million of debt, 8 million of which is a construction loan specifically for the construction of the plant. The rest of the...
-
Provide an example of an aggressive accounting practice. Why is this practice aggressive?
-
A stream of 5.00 wt% oleic acid in cottonseed oil enters an extraction unit at a rate of 100.0 kg/h. The unit operates as an equilibrium stage (the streams leaving the unit are in equilibrium) at...
-
The demand for a particular hydrogenated compound, S, is 5.00k mol/h. This chemical is synthesized in the gas-phase reaction A + H 2 = S. The reaction equilibrium constant at the reactor operating...
-
Terephthalic acid (TPA), a raw material in the manufacture of polyester fiber, film, and soft drink bottles, is synthesized from p-xylene (PX) in the process shown below. A fresh feed of pure liquid...
-
Why do you and your business need to be able to communicate well?
-
What are the basic criteria for effective messages?
-
What are the questions for analyzing a business communication situation?

Study smarter with the SolutionInn App


