Question: 6. Which reagent will accomplish this transformation? H (C) (A) H, Pd/BaSO, quinoline (B) NaBH. (C) LAIH (DI Na, NH, 57. What is the
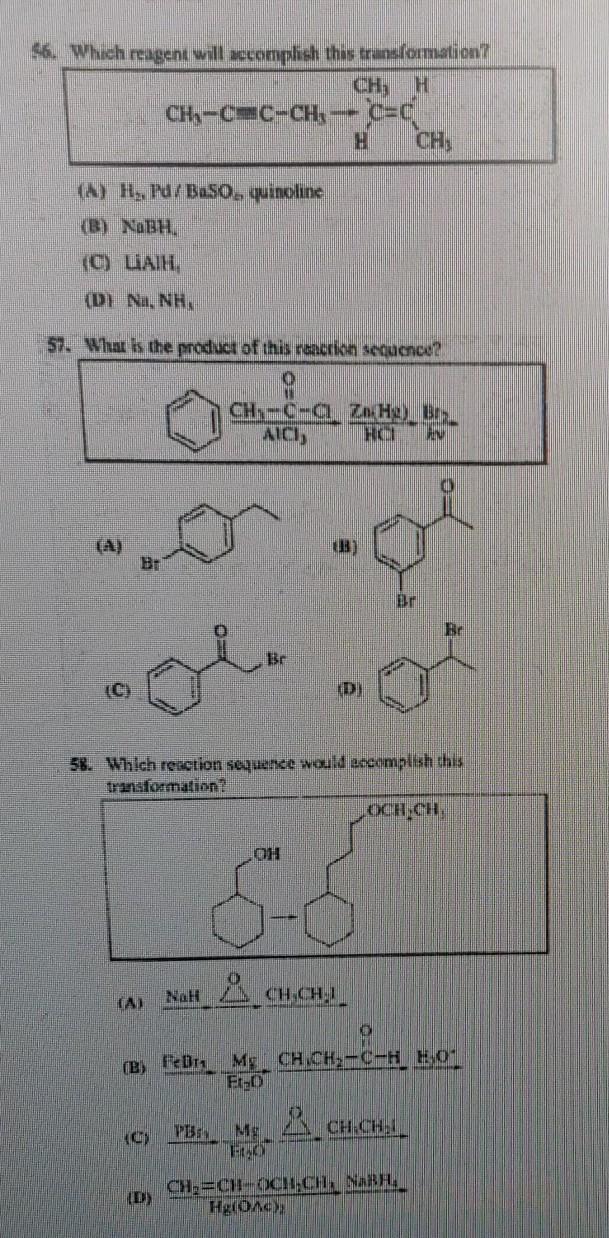
6. Which reagent will accomplish this transformation? H (C) (A) H, Pd/BaSO, quinoline (B) NaBH. (C) LAIH (DI Na, NH, 57. What is the product of this reaction sequence? (A) CH-C=C-CH G (D) CH-C-C AICI, NaH Br C=C H CH, ZnHg) Br H HO EN (B) 58. Which reaction sequence would accomplish this transformation? (D) CHICHI 13-1 LOCH,CH, O (B) FeBr My CH CH-C-H HO E1-D PB, MCH.CH. Br ERACY CH=CH-CHCH. NABH Hg(OAC);
Step by Step Solution
3.37 Rating (147 Votes )
There are 3 Steps involved in it

Get step-by-step solutions from verified subject matter experts


