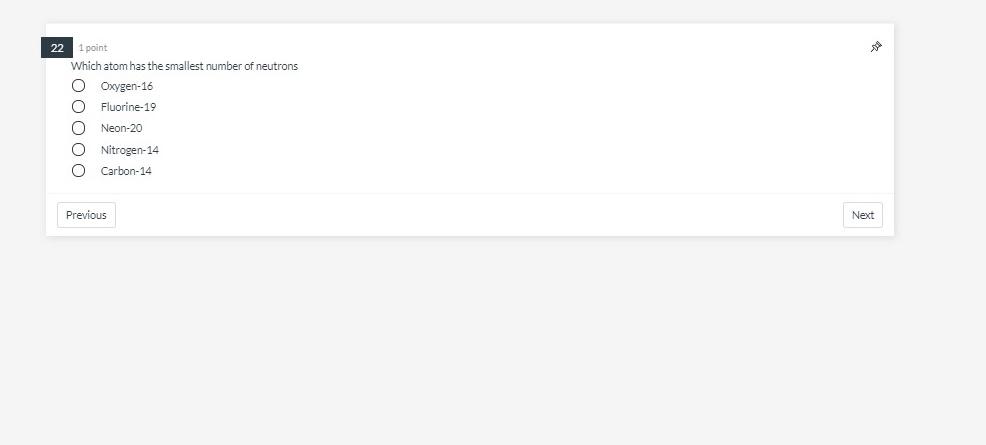
Question: 22 1 point Which atom has the smallest number of neutrons C Oxygen-16 O Fluorine-19 O Neon-20 C Nitrogen-14 O Carbon-14 Previous sa Next
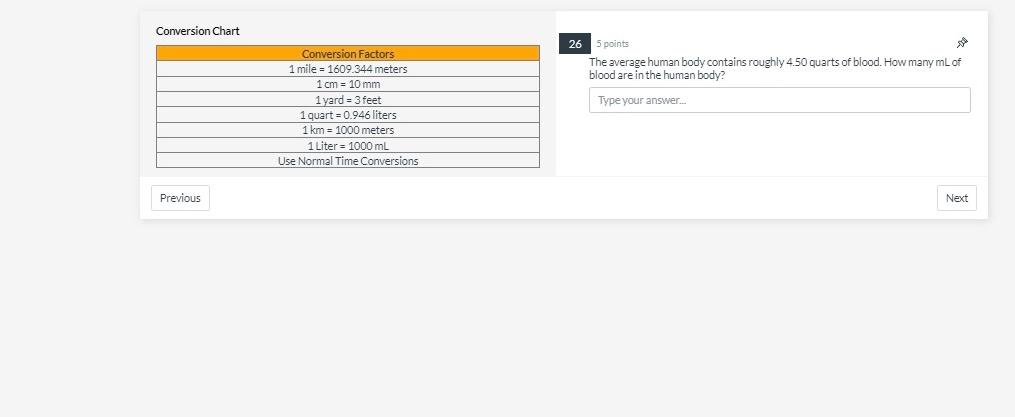
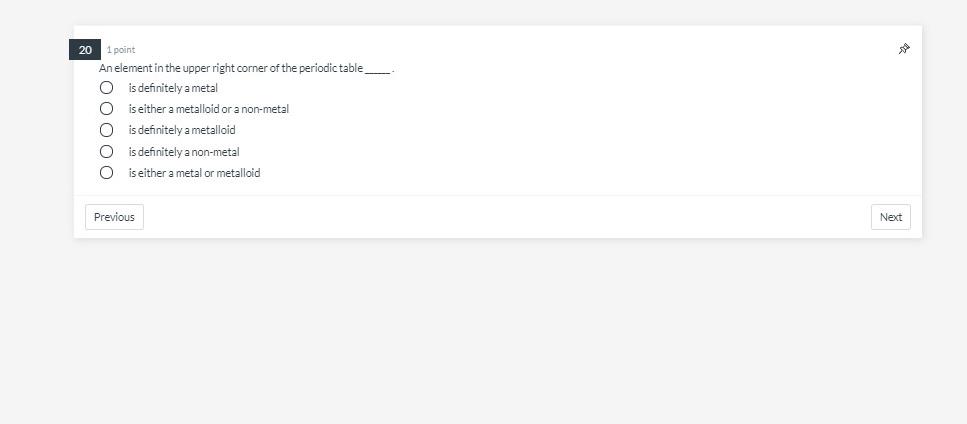
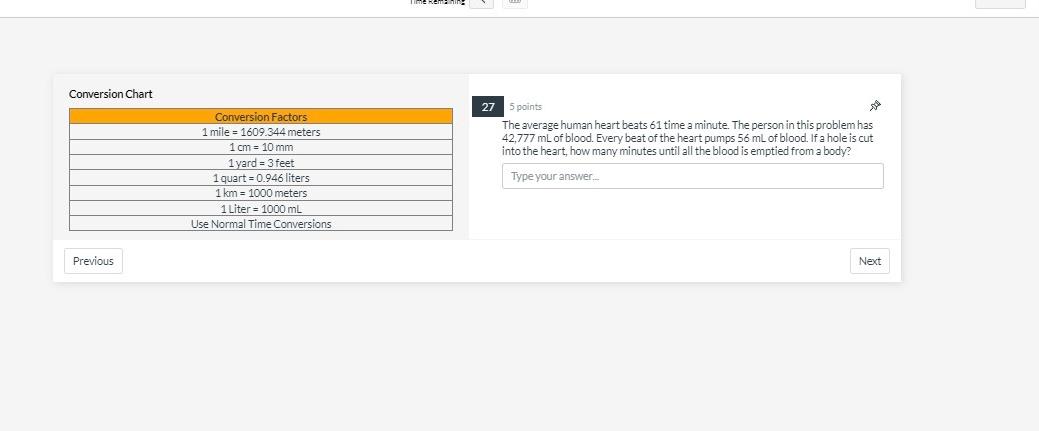
22 1 point Which atom has the smallest number of neutrons C Oxygen-16 O Fluorine-19 O Neon-20 C Nitrogen-14 O Carbon-14 Previous sa Next Conversion Chart Previous Conversion Factors 1 mile = 1609.344 meters 1 cm = 10 mm 1 yard - 3 feet 1 quart=0.946 liters 1 km 1000 meters 1 Liter = 1000 mL Use Normal Time Conversions 26 5 points: Jul The average human body contains roughly 4.50 quarts of blood. How many mL of blood are in the human body? Type your answer... Next 20 1 point An element in the upper right corner of the periodic table O is definitely a metal is either a metalloid or a non-metal is definitely a metalloid is definitely a non-metal is either a metal or metalloid 00000 Previous Next Conversion Chart Previous Conversion Factors 1 mile 1609.344 meters 1 cm = 10 mm 1yard = 3 feet 1 quart=0.946 liters 1 km = 1000 meters 1 Liter = 1000 mL Use Normal Time Conversions 27 5 points The average human heart beats 61 time a minute. The person in this problem has 42,777 mL of blood. Every beat of the heart pumps 56 mL of blood. If a hole is cut into the heart, how many minutes until all the blood is emptied from a body? Type your answer.... Next 12 1 point Which of the following has the same number of significant figures as the number 1.00310? 100 199.791 8.66 5.119 1x 108 Previous 42 Next
Step by Step Solution
3.40 Rating (163 Votes )
There are 3 Steps involved in it

Get step-by-step solutions from verified subject matter experts


