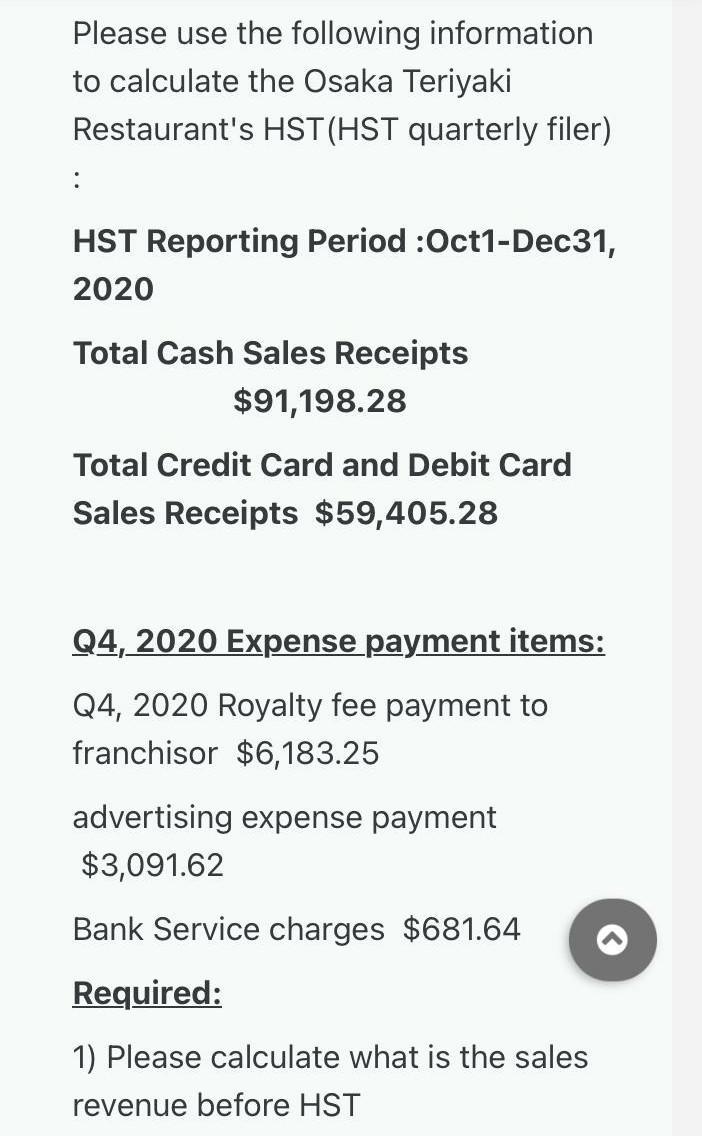
Please use the following information to calculate the Osaka Teriyaki Restaurant's HST (HST quarterly filer) :...
Fantastic news! We've Found the answer you've been seeking!
Question:

Transcribed Image Text:
Please use the following information to calculate the Osaka Teriyaki Restaurant's HST (HST quarterly filer) : HST Reporting Period :Oct1-Dec31, 2020 Total Cash Sales Receipts $91,198.28 Total Credit Card and Debit Card Sales Receipts $59,405.28 Q4, 2020 Expense payment items: Q4, 2020 Royalty fee payment to franchisor $6,183.25 advertising expense payment $3,091.62 Bank Service charges $681.64 Required: 1) Please calculate what is the sales revenue before HST Required: 1) Please calculate what is the sales revenue before HST 2) Please show me the breakdown calculation for HST payable to CRA at the end of the Q4, 2020. 3) When is the deadline to pay the Q4, 2020 HST 4) If the business already paid HST installment $17,000, how much is the final HST payable amount or refund amount? 5) If the Q4, 2020 Income Statement is generated, What is the net income for Q4, 2020? Please use the following information to calculate the Osaka Teriyaki Restaurant's HST (HST quarterly filer) : HST Reporting Period :Oct1-Dec31, 2020 Total Cash Sales Receipts $91,198.28 Total Credit Card and Debit Card Sales Receipts $59,405.28 Q4, 2020 Expense payment items: Q4, 2020 Royalty fee payment to franchisor $6,183.25 advertising expense payment $3,091.62 Bank Service charges $681.64 Required: 1) Please calculate what is the sales revenue before HST Required: 1) Please calculate what is the sales revenue before HST 2) Please show me the breakdown calculation for HST payable to CRA at the end of the Q4, 2020. 3) When is the deadline to pay the Q4, 2020 HST 4) If the business already paid HST installment $17,000, how much is the final HST payable amount or refund amount? 5) If the Q4, 2020 Income Statement is generated, What is the net income for Q4, 2020?
Expert Answer:
Answer rating: 100% (QA)
Ans1 Ans2 HST paid Bank Services are HST exempt Net ... View the full answer

Related Book For 

Posted Date:
Students also viewed these accounting questions
-
Use the following information to calculate ending balance in accounts receivable. (This is a tricky question. All the information is relevant. Set up T-accounts or a spreadsheet to help.) Accounts...
-
Use the following information to calculate the cost of goods sold for The Ellis Company for the month of June: Finished Goods Inventory: Beginning ................ $ 26,000 Ending ....................
-
Use the following information to calculate accounts receivable on July 31, 2018 and the amount of accounts receivable written off in 2018. Assume that all sales are oncredit: July 31 2018 2017...
-
What is the output of the following? A. 1223445 B. 2445 C. 22445 D. 223445 E. 2233445 F. None of the above. public class InitOrder { } {System.out.print("1"); } static System.out.print("2"); } public...
-
1. The city claimed that it benefited the public by allowing the bid change because it resulted in monetary savings for the city. Does this explanation justify the citys disregard of established...
-
1. Ladder in Static Equilibrium: A uniform ladder of mass M and length L is being used to paint your house. You rest the ladder against the frictionless front wall of your home, at an angle , from...
-
Consider a single-degree-of-freedom system with Coulomb damping (which offers a constant friction force, \(F_{c}\) ). Derive an expression for the force transmissibility when the mass is subjected to...
-
Using your results from Problem 6, (a) Calculate the probability that the project will be completed in 38 weeks. (b) Calculate the probability that the project will be completed in 42weeks. Most...
-
Explain how social media and related technology can be used to gather data and engage customers in the hotel and hospitality arena . Identify risks and gains. To complete your discussion, define what...
-
You will play the roles of a Freight Forwarder and a materials and distribution manager for Apotex Pharmaceuticals in Toronto. Your role is the materials and distribution manager for Apotex...
-
Which one of the following statements about starch and glycogen is correct? (a)Both starch and glycogen are found in plant cells. (b)Both starch and glycogen are found in animal cells. (c)Starch is...
-
Why is cost accumulation easier with a process costing system compared with a job costing system?
-
Profile the economy of Belgium: What is its GDP? What is its per capita income? How fast is its economy growing? What are its major exports and imports? Who are its major trading partners? Success in...
-
Euclid (300 B.C.) a. published a book on the theory of sound b. first person to investigate musical sounds on a scientific basis c. wrote a treatise called Introduction to Harmonics d. founder of...
-
Wind-induced vibration a. can cause failure of turbines and aircraft engines b. cause discomfort in human activity during metal cutting c. can cause wheels of locomotives to rise off the track d. can...
-
Provide examples of industries that use process costing.
-
Radiation properties and surface conditions for a four-zone enclosure with all sides are equal shown in the figure T = 500 K =0.7 T2 = 1000 K 2 =0.9 T3 800 K 3 =0.8 T4 = 900 K 4 = 0.6 Determine the...
-
Match the following. Answers may be used more than once: Measurement Method A. Amortized cost B. Equity method C. Acquisition method and consolidation D. Fair value method Reporting Method 1. Less...
-
How much does Reitmans report for trade and other receivables on January 28, 2012? Why do you think the amount of trade and other receivables is so small?
-
What is goodwill ? How does it arise? How much goodwill did TELUS report on December 31, 2011? How does TELUS amortize its goodwill ? How much new goodwill did TELUS add in 2011? Is it possible that...
-
The following information is provided for Clova Ltd. (Clova): Required: a. Calculate ending inventory on December 31, 2017 and 2018, and cost of sales and gross margin for the years ended December...
-
Show that for steady state flow process, \(\Delta H=Q-W_{S}\).
-
What is reaction coordinate? What is its significance in chemical reaction?
-
An ideal gas \(\left(C_{P}=5, C_{V}=3 ight)\) is changed from \(1 \mathrm{~atm}\) and \(22.4 \mathrm{~m}^{3}\) to \(10 \mathrm{~atm}\) and 2.24 \(\mathrm{m}^{3}\) by the reversible process of heating...

Study smarter with the SolutionInn App


